BMJ 2001;323:81-85 ( 14 July )
Papers
![]()
Reduced osmolarity oral rehydration solution for treating
dehydration due to diarrhoea in children: systematic review
Seokyung Hahn
a Medical and Pharmaceutical
Statistics Research Unit, University of Reading, Reading RG6 6FN, b Department
of Paediatrics, Seoul National University Children's Hospital, Seoul
110-774, South Korea, c Effective Health Care Alliance
Programme, International Health Division, Liverpool School of Tropical
Medicine, Liverpool L3 5QA
Correspondence to: S Hahn, Department of Health Sciences and
Clinical Evaluation, University of York, York YO10 5DD [log in to unmask]
|
|
Abstract |
Objectives: To compare reduced osmolarity oral
rehydration solution with standard World Health Organization oral
rehydration solution in children with acute diarrhoea.
Design: Systematic review of
randomised controlled trials.
Studies: 15 randomised controlled
trials including 2397 randomised patients.
Outcomes: The primary outcome was
unscheduled intravenous infusion; secondary outcomes were stool
output, vomiting, and hyponatraemia.
Results: In a meta-analysis of
nine trials for the primary outcome, reduced osmolarity rehydration
solution was associated with fewer unscheduled intravenous infusions
compared with standard WHO rehydration solution (odds ratio
0.61, 95% confidence interval 0.47 to 0.81). Three trials
reported that no patients required unscheduled intravenous infusion.
Trials reporting secondary outcomes suggested that in the reduced
osmolarity rehydration solution group, stool output was lower
(standardised mean difference in the log scale ![]() 0.214 (95% confidence interval
0.214 (95% confidence interval ![]() 0.305 to
0.305 to ![]() 0.123; 13 trials)
and vomiting was less frequent (odds ratio 0.71, 0.55 to
0.92; six trials). Six trials sought presence of hyponatraemia, with
events in three studies, but no significant difference between the
two arms.
0.123; 13 trials)
and vomiting was less frequent (odds ratio 0.71, 0.55 to
0.92; six trials). Six trials sought presence of hyponatraemia, with
events in three studies, but no significant difference between the
two arms.
Conclusion: In children admitted
to hospital with dehydration associated with diarrhoea, reduced
osmolarity rehydration solution is associated with reduced need for
unscheduled intravenous infusions, lower stool volume, and less
vomiting compared with standard WHO rehydration solution.
|
What is already known on this topic What this study adds Reduced osmolarity rehydration solution
also reduced stool output and vomiting No difference was found in rates of hyponatraemia
|
|
|
Introduction |
Diarrhoea remains a leading cause of childhood death in developing
countries. The main complication is dehydration, which until the
early 1960s was treated with intravenous infusion. Solutions of oral
rehydration salts are now the main treatment and are particularly useful
when intravenous fluids are in short supply, health services are
basic, and there is a shortage of skilled staff.1 The
combination of salt and sugar probably enhances absorption of fluid
because sodium and glucose transport in the small intestine are
coupled; glucose promotes absorption of both sodium ions and water.2 Oral
rehydration salts have proved both safe and effective worldwide in
hospital settings and are now widely used in the home to prevent dehydration.
3 4
For more than two decades, the World Health Organization has
recommended a standard formulation of glucose based oral rehydration solution
with 90 mmol/l of sodium and 111 mmol/l of glucose and a
total osmolarity of 311 mmol/l. It remains unclear however, whether
this is the optimum sodium concentration. Some studies have found
patients with blood sodium concentrations above the normal level of
150 mmol/l.5
Laboratory work suggests that lower concentrations of sodium and
glucose enhance solute induced water absorption. 6 7
We conducted a systematic review of all relevant randomised
controlled trials comparing the effects of reduced osmolarity and
standard WHO oral rehydration solutions. We confined the review to
children, as they are most vulnerable to dehydration and electrolyte imbalance
from diarrhoea.
|
|
Methods |
Study inclusion and characteristics
We included only randomised controlled trials, defined as a trial in
which the subjects were assigned prospectively to one of two or more
interventions by random allocation. This excludes quasirandomised
designs. Patients included were children with acute diarrhoea for less
than 5 days who were treated either by reduced osmolarity oral
rehydration solution (total osmolarity ![]() 250 mmol/l with reduced sodium) or by standard WHO oral
rehydration solution (sodium 90 mmol/l, glucose 111mmol/l,
total osmolarity 311 mmol/l).
250 mmol/l with reduced sodium) or by standard WHO oral
rehydration solution (sodium 90 mmol/l, glucose 111mmol/l,
total osmolarity 311 mmol/l).
The primary outcome was specified as the
need for unscheduled intravenous infusion during the course of treatment. This
was defined as clinical requirement for intravenous infusion after
oral rehydration had been started. We chose unscheduled intravenous infusion
as the primary outcome because it is a pragmatic outcome that is
relevant to health providers and represents failed oral treatment.
Other outcomes were stool output, vomiting during rehydration, and
presence of hyponatraemia (serum sodium concentration <130mmol/l) during
follow up.
Search strategy
We searched the following databases for published clinical trials:
Medline (1966 to June 2001); Embase (1988 to May 2001);
Cochrane controlled trials register in the Cochrane
Library (Issue 2, 2001); and Current Contents (June
2001). We used child, diarrhoea, fluid therapy, oral rehydration,
osmolar, and rehydration solutions as search terms. We also checked
the citations of existing reviews and trial reports. For unpublished
data and ongoing trials, we contacted current researchers and key
agencies, including the WHO, the Centers for Disease Control, Atlanta,
and the International Centre for Diarrhoeal Disease Research, Bangladesh.
Data extraction and synthesis
We used the standard methods of the Cochrane infectious diseases
group to prepare the protocol, apply inclusion criteria, assess
quality, and extract data. We assessed quality by adequacy of
concealment of allocation, generation of allocation sequence,
blinding, and follow up of patients. The first two authors independently
extracted data on relevant outcome measures using a standardised
data abstraction form, and any disagreements were resolved by discussion.
We used the Mantel-Haenszel odds ratio for
binary outcomes. The odds ratios were not estimated when neither intervention
group found any event. We used the standardised mean difference for
continuous outcomes. We combined studies using a fixed effect method
based on a weighted average of the results with weights proportional
to the inverse of the variance.8 For all
estimates, we calculated 95% confidence intervals. We tested
statistical heterogeneity using ![]() 2 tests, with a P value
2 tests, with a P value ![]() 0.1 indicating significance. We had prespecified potential
sources of heterogeneity for analysis.9 We
examined publication bias using a funnel plot and a regression
approach to assess asymmetry of the plot.10 We also
did a sensitivity analysis to assess the effect of adequacy of
concealment of allocation.
0.1 indicating significance. We had prespecified potential
sources of heterogeneity for analysis.9 We
examined publication bias using a funnel plot and a regression
approach to assess asymmetry of the plot.10 We also
did a sensitivity analysis to assess the effect of adequacy of
concealment of allocation.
|
|
Results |
Profile of studies
Of 41 identified studies, six were not randomised trials, eight
had not used glucose based reduced osmolarity rehydration solution,
six had not used standard WHO rehydration solution, two were in
adults, and two did not report on any of the relevant outcomes.
Seventeen studies in 16 published reports met the inclusion criteria.11-26 One
paper reported on two trials,24 one in
the United States and one in Panama, and we present these as
separate studies. We contacted the authors of three papers, 22 25 26 as we
judged these were three reports of the same trial. As we have not
received a response, we included only the paper with the largest
number of patients.22
Description and quality of studies
Details of the 15 trials included in the analysis and their
patient characteristics are available on the BMJ's
website. Included studies were from Egypt (three studies),
Bangladesh (three), Mexico (one), Colombia (one), India (three),
Panama (one), and the United States (one). Two other studies were
multicentre trials; one was conducted in Brazil, India, Mexico, and
Peru and the other in Bangladesh, Brazil, India, Peru, and Vietnam.
Three trials included children with
cholera. 12
14 21 Children
were aged 1-36 months in all trials except one, which included children
up to 5 years old.21
All children had some degree of clinical dehydration. One trial
treated all children on day 1 with intravenous infusion, and
those with a stool output of 80 ml/kg/24 h were then
randomised.11
Five trials included children with severe dehydration. 14 21 23 24 Five
trials included malnourished children. 12 15 20-22 The
number of breastfed children was reported in eight trials. 11-15 18 21 24 Fully
weaned children were included in one trial.16
We deviated slightly from the osmolarity
definitions in our refereed protocol published in the Cochrane Library.9 We defined
reduced osmolarity as <250 mmol/l, but some studies defined reduced
osmolarity as higher than this, and we therefore extended our limit
to <270 mmol/l. We also included two studies that used a WHO oral
rehydration solution with a total osmolarity of 331 mmol/l rather
than 311 mmol/l but with the same sodium and glucose combination.23 All
but three trials used a glucose based reduced osmolarity rehydration solution;
one used sucrose,13
another maltodextrin,17
and the third used L-alanine with glucose.11
|
Nine trials reported
methods that assured adequate concealment of allocation. Seven studies stated
they were double blinded, and eight did not mention blinding.
Quantitative data synthesis
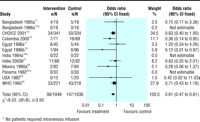
Figures 1-4 show the
meta-analyses for the four outcomes. Information on the primary
outcome (unscheduled intravenous infusion) was available in
12 trials (n=2085), but in three of these no patients in either
group required infusion. In the meta-analysis of nine trials, the
need for intravenous infusion was significantly reduced for
participants who received reduced osmolarity rehydration solution
compared with those receiving WHO rehydration solution (odds ratio
0.61, 95% confidence interval 0.47 to 0.81).
|
|
|
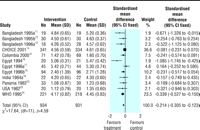
Thirteen trials reported
stool output during rehydration. These trials measured stool output in various
ways using different units. We therefore used the standardised mean
difference to analyse these data. Since the stool output in
diarrhoeal disease showed a positive skewed distribution with
clinical improvement, we used a log-normal approximation. The pooled
standardised mean difference in the log scale is ![]() 0.214 (95% confidence interval
0.214 (95% confidence interval ![]() 0.305 to
0.305 to ![]() 0.123),
which suggests that the reduced osmolarity rehydration solution
resulted in significantly less stool output than the WHO solution.
Data from one trial19
were not combined with the others in the meta-analysis because this
trial measured stool output for much longer than the others (see BMJ's website for details of
results).
0.123),
which suggests that the reduced osmolarity rehydration solution
resulted in significantly less stool output than the WHO solution.
Data from one trial19
were not combined with the others in the meta-analysis because this
trial measured stool output for much longer than the others (see BMJ's website for details of
results).
Six trials reported on vomiting during
rehydration. The tendency was for fewer patients to vomit in the reduced
osmolarity rehydration solution group (odds ratio
0.71, 0.55 to 0.92).
Six trials reported on hyponatraemia. In
three of these six trials, no patients had hyponatraemia. The meta-analysis of
the three trials in which participants developed hyponatraemia
showed no significant difference between the groups (odds ratio
1.45, 0.93 to 2.26). We found no evidence of statistical
heterogeneity of treatment effect for any of the four outcomes.
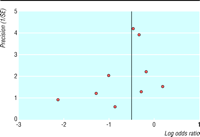
The funnel plot of the primary outcome
showed no significant evidence of publication bias (fig 5). The regression
method used to assess funnel plot asymmetry gave an intercept of ![]() 0.72 with a
P value of 0.29. A sensitivity analysis restricted to studies with
clear evidence of adequate concealment of allocation produced results
that did not differ greatly from those of the full meta-analysis. For
example, the pooled odds ratios of the seven trials for the primary
outcome with adequate concealment of allocation was 0.61 (0.46 to
0.82).
0.72 with a
P value of 0.29. A sensitivity analysis restricted to studies with
clear evidence of adequate concealment of allocation produced results
that did not differ greatly from those of the full meta-analysis. For
example, the pooled odds ratios of the seven trials for the primary
outcome with adequate concealment of allocation was 0.61 (0.46 to
0.82).
|
|
Discussion |
We found that reduced osmolarity rehydration solution was more
effective than standard WHO rehydration solution in first line treatment
of children with diarrhoea. It reduced the need for unscheduled
intravenous infusion, stool output during rehydration, and the
number of patients with vomiting during oral rehydration treatment.
The reduced osmolarity rehydration solution did not seem to increase
the risk of developing hyponatraemia compared with the standard WHO
solution, although we cannot exclude this possibility.
We examined trials of oral rehydration salts in children admitted
to hospital with dehydration because of diarrhoea. The trials do not
provide any direct evidence for or against use of oral rehydration
solutions at home to prevent dehydration; nor do they provide any
direct evidence that reduced osmolarity solutions are more effective
in preventing dehydration in the home. Oral rehydration solutions
are widely used to prevent dehydration, and further research is
therefore needed in this area.
|
Choice of primary outcome
We stand by our selection of unscheduled intravenous infusion rather
than volume of diarrhoea as the primary outcome.9 Some
specialists consider that volume of diarrhoea is more appropriate, probably
because it reflects the animal and human perfusion experiments that
provide part of the rationale for a reduced osmolarity rehydration solution.
Unscheduled intravenous infusion is pragmatic; it provides a measure
of failed oral rehydration and has implications for healthcare
resources. For these reasons, we preserved this as the primary outcome.
When we reviewed the studies for
inclusion, most trials reported unscheduled intravenous infusion in the details
of trial implementation. As this was identified as our primary
outcome, we sought out these data and presented them as the primary
analysis. We believe that the analysis shows a clear effect. Our
approach highlights the value of paying careful attention to the
protocol for a systematic review before examining the trials and
illustrates how non-specialist viewpoints can help obtain practical
and useful answers from a meta-analysis.
Cholera
We intended to examine treatment effects in patients with and
without cholera.9
A Cochrane review of rice based rehydration compared with glucose
oral rehydration solution showed that rice water was associated with
lower stool volumes in cholera patients but not in diarrhoea from
other causes.27
The available data were, however, insufficient. Three studies included
cholera patients, 12
14 21 but
separate data for cholera patients were not available. We excluded
two studies in patients with cholera because they were in adults. 28 29 Any
recommendation for rehydration treatment for adults with cholera
will need to take into account these and any other trials found
through careful systematic searching.
Patients with cholera have severe loss of
electrolytes. It is unclear, therefore, whether reduced osmolarity rehydration
solution would be more effective than standard WHO rehydration
solution in these patients. The reduced osmolarity solution could
increase the risk of hyponatraemia and result in adverse clinical
events
Implications
Our study suggests that reduced osmolarity rehydration solution
should replace the WHO solution as the standard treatment for
dehydration caused by diarrhoea. Policymakers and clinicians may,
however, consider that the risk of hyponatraemia in patients with
cholera outweighs the advantages of a reduced osmolarity solution.
One option would be to provide standard WHO rehydration solution for
people with suspected cholera or in areas where cholera is
prevalent. This is likely to be a logistical problem in areas where
diarrhoea is common and coexists with cholera. The single formula
sachet aids implementation of this lifesaving intervention.
Providing different formulations complicates distribution and may
impair the effective delivery of any oral rehydration solution to
children. If policymakers decide not to use reduced osmolarity
solution in areas where cholera is common, they must conduct a
randomised controlled trial of the two treatments in children with
cholera to determine whether the decision is correct.
|
|
Acknowledgments |
This review will be maintained in the
Cochrane Library.
We thank Dr Olivier Fontaine (WHO) for support throughout the
process; and Dr Sheila Bird, Medical Research Council, Cambridge, for
her comments.
Contributors: SH and YK wrote the protocol, extracted, analysed
and interpreted the data, and drafted the paper. PG advised on inclusion
criteria and outcomes for the protocol, quality assessment, and analysis, and
helped write the review. PG is the guarantor.
|
|
Footnotes |
Funding : The study is supported by WHO and the Cochrane
Infectious Diseases Group is supported by the Department for International
Development.
Competing interests: None declared.
![]() Tables giving further details of the included trials are
available on the BMJ's website
Tables giving further details of the included trials are
available on the BMJ's website
|
|
References |
|
1. |
Almroth S, Latham MC. Rational home
management of diarrhoea. Lancet
1995; 345: 709-711 |
|
2. |
Fordtran JS, Rector FC, Carter NW. The
mechanisms of sodium absorption in the human small intestine. J Clin Invest 1968; 47: 884-900 |
|
3. |
Mahalanabis M, Choudfuri AB, Bagchi NG.
Oral fluid therapy of cholera among Bangladesh refugees. Johns Hopkins Med J 1973; 132: 197-205 |
|
4. |
Grant JP. The state of the world's children 1982-1983. New York:
UNICEF, 1983. |
|
5. |
Finberg L. Hypernatremic (hypertonic)
dehydration in infants. N Engl J Med
1973; 289: 196-198 |
|
6. |
Farthing MJ. History and rationale of
oral rehydration and recent developments in formulating an optimal solution. Drugs 1988; 36(suppl 4): 80-90 |
|
7. |
Hunt JB, Elliotte EJ, Fairclough PD,
Clark ML, Farthing MJ. Water and solute absorption from hypotonic
glucose-electrolyte solutions in human jejunum. Gut 1992; 33: 479-483 |
|
8. |
Sutton AJ, Abrams KR, Jones DR, Sheldon
TA, Song F. Fixed effects methods for combining study estimates. In: Methods for meta-analysis in medical research.
Chichester: Wiley, 2000. |
|
9. |
Kim Y, Hahn S, Garner P. Reduced
osmolarity oral rehydration solution for treating diarrhoea-associated
dehydration in children (protocol for a Cochrane review). In: Cochrane
Collaboration,ed. Cochrane Library.
Issue 4. Oxford: Update Software, 2000. |
|
10. |
Egger M, Davey Smith G, Schneider M,
Minder C. Bias in meta-analyses detected by a simple graphical test. BMJ 1997; 315: 629-634 |
|
11. |
Sarker SA, Majid N, Mahalanabis D.
Alanine- and glucose-based hypo-osmolar oral rehydration solution in infants
with persistent diarrhoea: a controlled trial. Acta Paediatr 1995; 84: 775-780 |
|
12. |
Mahalanabis D, Faruque ASG, Hoque SS,
Faruque SM. Hypotonic oral rehydration solution in acute diarrhoea: a
controlled clinical trial. Acta Paediatr
1995; 84: 289-293 |
|
13. |
Faruque ASG, Mahalanabis D, Hamadani J,
Hoque SS. Hypo-osmolar sucrose oral rehydration solution in acute diarrhoea:
a pilot study. Acta Paediatr
1996; 85: 1247-1248 |
|
14. |
CHOICE study group. Multicenter
randomized double blind clinical trial to evaluate the efficacy and safety of
a reduced osmolarity oral rehydration salts solution in children with acute
watery diarrhoea. Pediatrics
2000; 107: 613-618 |
|
15. |
Bernal C, Velaquez C, Garcia G, Uribe G,
Palacio C. Oral hydration with a low osmolarity solution in children
dehydrated by diarrheic diseases. A controlled clinical study. Saludarte 2000; 1: 6-23 |
|
16. |
El-Mougi M, El-Akkad N, Hendawi A,
Hassan M, Amer A, Fontaine O, Pierce NF. Is a low-osmolarity ORS solution
more efficacious than standard WHO ORS solution? J Pediatr Gastroenterol Nutr 1994; 19(1): 83-86 |
|
17. |
El-Mougi M, Hendawi A, Koura H, Hegazi
E, Fontaine O, Pierce NF. Efficacy of standard glucose-based and reduced
osmolarity maltodextrin-based oral rehydration solution: effect of sugar
malabsorption. Bull World Health Organ
1996; 74(5): 471-477 |
|
18. |
Santosham M, Fayad I, Zikri MA, Hussein
A, Amponsah A, Duggan C, et al. A double-blind clinical trial comparing World
Health Organization oral rehydration solution with a reduced osmolarity
solution containing equal amounts of sodium and glucose. J Pediatr 1996; 128(1): 45-51 |
|
19. |
Bhargava SK, Sachdev HPS, Das Gupta B,
Daral TS, Singh HP, Mohan M. Oral rehydration of neonates and young infants
with dehydration diarrhea: comparison of low and standard sodium content in
oral rehydration solutions. J Pediatr
Gastroenterol Nutr 1984; 3(4): 500-505 |
|
20. |
Dutta P, Mitra U, Dutta S, Manna B,
Chatterjee MK, De A, et al. Hypo-osmolar oral rehydration salts solution in
dehydrating persistent diarrhoea in children: double-blind, randomized,
controlled clinical trial. Acta Paediatr
2000; 89: 411-416 |
|
21. |
Alam S, Afzal K, Maheshwari M, Shukla I.
Controlled trial of hypo-osmolar versus World Health Organization oral
rehydration solution. Indian Pediatr
2000; 37: 952-959 |
|
22. |
Moreno-Sanchez H, Velasques-Jones L,
Becerra FC, Faure A, Maulen I, de Leon M, et al. A comparative study on two
oral rehydration solutions (ORS) containing 60 or 90 mmol/L of
sodium and of different osmolalities. Bol
Med Hosp Infant Mex 1990; 47(9): 630-635 |
|
23. |
Santosham M, Daum RS, Dillman L,
Rodriguez JL, Luque S, Russel R, et al. Oral rehydration therapy of infantile
diarrhea. A controlled study of well-nourished children hospitalized in the
United States and Panama. New Engl J Med
1982; 306(18): 1070-1076 |
|
24. |
World Health Organization. International
study group on reduced-osmolarity ORS solutions. Multicentre evaluation of
reduced-osmolarity oral rehydration salts solution. Lancet 1995; 345: 282-285 |
|
25. |
Martinez-Pantaleon O, Faure-Vilchis A,
Gomez-Najera RI, Hernandez-Lopez M, Velasquez-Jones L. Comparative study of
oral rehydration solutions containing either 90 or 60 millimoles of
sodium per liter. Bol Med Hosp Infant Mex
1998; 45(12): 817-822 |
|
26. |
Velasquez-Jones L, Becerra FC, Faure A,
de Leon M, Moreno H, Maulen I, et al. Clinical experience in Mexico with a
new oral rehydration solution with lower osmolality. Clin Ther 1990; 2(suppl A): 95-103 |
|
27. |
Fontaine O, Gore S, Pierce NF.
Rice-based oral rehydration solution for treating diarrhoea. Cochrane Database Syst Rev 2000; (2):
CD001264 |
|
28. |
Faruque ASG, Mahalanabis D, Hamadani JD,
Zetterstrom R. Reduced osmolarity oral rehydration salt in cholera. Scand J Infect Dis 1996; 28: 87-90 |
|
29. |
Alam NH, Majumder RN, Fuchs GJ, CHOICE
Study Group. Efficacy and safety of oral rehydration solution with reduced
osmolarity in adults with cholera: a randomised double-blinded clinical
trial. Lancet 1999; 354: 296-299 |
(Accepted 27 June 2001)
Edward E.
Rylander, M.D.
Diplomat American
Board of Family Practice.
Diplomat American
Board of Palliative Medicine.