
Review Article
MEDICAL PROGRESS
Volume 341 Number 11 (September 9, 1999) New
England Journal of Medicine 1999;341:815-26.
ANTHRAX
TERRY C. DIXON, B.S., MATTHEW MESELSON, PH.D., JEANNE GUILLEMIN, PH.D., AND PHILIP C.
HANNA, PH.D.
From the Department of Microbiology, Duke
University Medical Center, Durham, N.C. (T.C.D., P.C.H.); the Department of
Molecular and Cellular Biology, Harvard University, Cambridge, Mass. (M.M.);
the Department of Sociology, Boston College, Chestnut Hill, Mass. (J.G.); and
the Department of Microbiology and Immunology, University of Michigan Medical
School, Ann Arbor (P.C.H.). Address reprint requests to Dr. Hanna at 1150 W
Medical, 5641 MS II, Department of Microbiology and Immunology, University of
Michigan Medical School, Ann Arbor, MI 48104, or at [log in to unmask].
Copyright©1999, Massachusetts Medical Society.
All rights reserved
ANTHRAX is an often fatal bacterial infection
that occurs when Bacillus anthracis endospores
enter the body through abrasions in the skin or by inhalation or ingestion. 1 It is a zoonosis to
which most mammals, especially grazing herbivores, are considered susceptible.
Human infections result from contact with contaminated animals or animal
products, and there are no known cases of human-to-human transmission. Human
anthrax is not common, and only one of us has seen a case. Cutaneous anthrax,
the most common form, is usually curable. A small percentage of cutaneous infections
become systemic, and these can be fatal. Systemic infection resulting from
inhalation of the organism has a mortality rate approaching 100 percent, with
death usually occurring within a few days after the onset of symptoms. 2 The rate of mortality
among persons with infection resulting from ingestion is variable, depending on
the outbreak, but it may also approach 100 percent. Whatever the portal of
entry, systemic anthrax involves massive bacteremia and toxemia with
nondescript initial symptoms until the onset of hypotension, shock, and sudden
death. Manifestations of advanced disease, including shock and sudden death,
are believed to result from the action of the exotoxin complex secreted by
anthrax bacilli. 1,3 The efficacy of therapy, if initiated during the incubation
period, and the rapid course of the disease once symptoms appear make early
intervention an absolute necessity. Inglesby et al. have provided a description
of the policies and strategies for dealing with anthrax as a biologic weapon. 4 The goal of this article
is to familiarize physicians with the current understanding of the
pathogenesis, diagnosis, prevention, and treatment of anthrax.
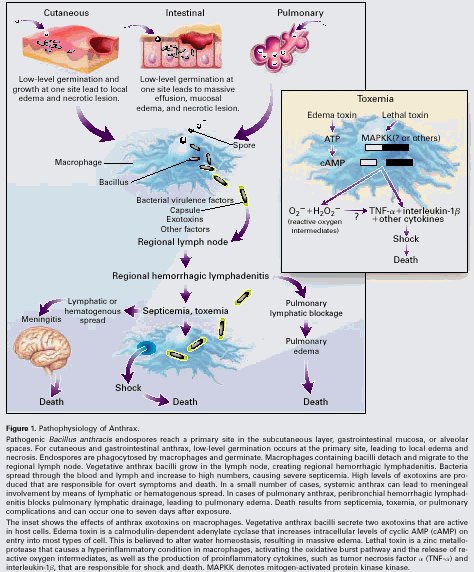
PATHOGENESIS
Anthrax infections are initiated by endospores
of B. anthracis, a gram-positive
soil organism. Anthrax endospores do not divide, have no measurable metabolism,
and are resistant to drying, heat, ultraviolet light, gamma radiation, and many
disinfectants. 5 In some types of soil, anthrax spores can remain dormant for
decades. Their hardiness and dormancy have allowed anthrax spores to be
developed as biologic weapons by a number of nations, although their only known
use in war was by the Japanese army in Manchuria in the 1940s. 6 All known anthrax
virulence genes are expressed by the vegetative form of B. anthracis that results from the
germination of spores within the body. The course of infection and clinical
manifestations are depicted in Figure 1.
Endospores introduced into the body by abrasion, inhalation, or ingestion are
phagocytosed by macrophages and carried to regional lymph nodes. Endospores
germinate inside the macrophages and become vegetative bacteria 7,8 ; the vegetative
bacteria are then released from the macrophages, multiply in the lymphatic
system, and enter the bloodstream, until there are as many as 107 to
108 organisms per milliliter of blood, causing massive septicemia.
Once they have been released from the macrophages, there is no evidence that an
immune response is initiated against vegetative bacilli. Anthrax bacilli express
virulence factors, including toxin and capsule. 1 The resulting toxemia
has systemic effects that lead to the death of the host. The major virulence
factors of B. anthracis are
encoded on two virulence plasmids, pXO1 and pXO2. The toxin-bearing plasmid,
pXO1, is 184.5 kilobase pairs (kbp) in size and codes for the genes that make
up the secreted exotoxins. The toxin-gene complex is composed of protective
antigen, lethal factor, and edema factor. 9 The three exotoxin
components combine to form two binary toxins. Edema toxin consists of edema
factor, which is a calmodulin-dependent adenylate cyclase, 10,11 and protective antigen,
the binding moiety that permits entry of the toxin into the host cell.
Increased cellular levels of cyclic AMP upset water homeostasis and are
believed to be responsible for the massive edema seen in cutaneous anthrax.
Edema toxin inhibits neutrophil. function in vitro, 12 and neutrophil function
is impaired in patients with cutaneous anthrax infection. 13 Lethal toxin consists of
lethal factor, which is a zinc metalloprotease 14-16 that inactivates
mitogen-activated protein kinase kinase in vitro, 17,18 and protective antigen,
which acts as the binding domain. Lethal toxin stimulates the macrophages to
release tumor necrosis factor a and
interleukin-1b, which are partly
responsible for sudden death in systemic anthrax (Fig 1, inset). The smaller capsule-bearing plasmid, pXO2,
is 95.3 kbp in size and codes for three genes (capB,
capC, and capA)
involved in the synthesis of the polyglutamyl capsule. 20 The exotoxins are
thought to inhibit the immune response mounted against infection, whereas the
capsule inhibits phagocytosis of vegetative anthrax bacilli. The expression of
all known major virulence factors is regulated by host-specific factors such as
elevated temperature and carbon dioxide concentration, and by the presence of
serum components. 21,22 Regulation of the expression of the toxin and capsule genes is
mediated by the transcriptional activator AtxA, whose activity appears to be
affected by the previously mentioned environmental conditions. 23-25 Expression of the
capsule gene is also controlled by its own transcriptional regulator, AcpA. 26 Both plasmids are
required for full virulence; the loss of either one results in an attenuated
strain. Historically, bacterial strains for anthrax vaccine were made by
rendering virulent strains free of one or both plasmids. Pasteur, an avirulent
pXO2-carrying strain, is encapsulated but does not express exotoxin components.
1 Sterne,
an attenuated strain that carries pXO1, can synthesize exotoxin components but
does not have a capsule. 1
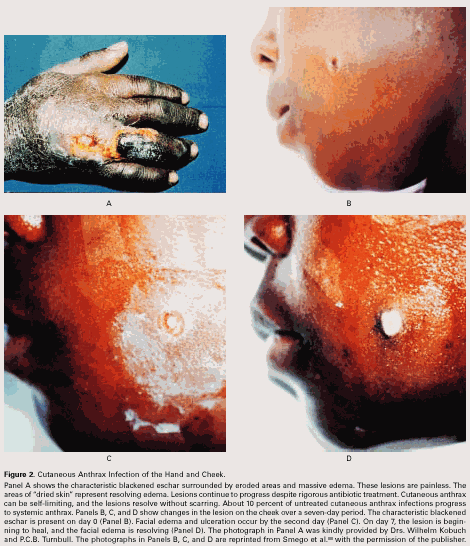
CLINICAL MANIFESTATIONS
Cutaneous Anthrax
Cutaneous anthrax accounts for 95 percent of all
anthrax infections in the United States. 27-30 The name anthrax (from
the Greek for coal) refers to the typical black eschar that is seen on affected
areas (Fig 2). Patients often
have a history of occupational contact with animals or animal products. The
most common areas of exposure are the head, neck, and extremities, although any
area can be involved. Pathogenic endospores are introduced subcutaneously
through a cut or abrasion. There are a few case reports of transmission by
insect bites, presumably after the insect fed on an infected carcass. 31,32 The primary skin lesion
is usually a nondescript, painless, pruritic papule that appears three to five
days after the introduction of endospores. In 24 to 36 hours, the lesion forms
a vesicle that undergoes central necrosis and drying, leaving a characteristic
black eschar surrounded by edema and a number of purplish vesicles. The edema
is usually more extensive on the head or neck than on the trunk or extremities.
33 The
common description malignant pustule is actually a misnomer, because the
cutaneous lesion is not purulent and is characteristically painless. A painful,
pustular eschar in a febrile patient indicates a secondary infection, most
often with staphylococcus or streptococcus. 34 Although cutaneous
anthrax can be self-limiting, antibiotic treatment is recommended. Lesions
resolve without complications or scarring in 80 to 90 percent of cases.
Malignant edema is a rare complication characterized by severe edema,
induration, multiple bullae, and symptoms of shock. 35,36 Malignant edema
involving the neck and thoracic region often leads to breathing difficulties
that require corticosteroid therapy or intubation. A few cases have been
reported of temporal arteritis associated with cutaneous anthrax infection and
of corneal scarring from palpebral cutaneous anthrax. 37,38 Histologic examination
of anthrax skin lesions shows necrosis and massive edema with lymphocytic infiltrates.
There is no liquefaction or abscess formation, indicating that the lesions are
not suppurative. Focal points of hemorrhage are evident, with some thrombosis. 39 Gram's staining reveals
bacilli in the subcutaneous tissue. 39
Gastrointestinal and Oropharyngeal Anthrax
Gastrointestinal anthrax, which can be fatal,
has not been reported in the United States. The symptoms appear two to five
days after the ingestion of endospore-contaminated meat from diseased animals. 40 Therefore, multiple
cases can occur within individual households. 40,41 An unusually prolonged
outbreak was attributed to the consumption of stored meat products. 42 It is presumed that
bacterial inoculation takes place at a breach in the mucosal lining, but
exactly where the endospores germinate is unknown. On pathological examination,
bacilli can be seen microscopically in the mucosal and submucosal lymphatic
tissue, and there is gross evidence of mesenteric lymphadenitis. 43 Ulceration is always
seen. It is not known whether ulceration occurs only at sites of bacterial
infection or is distributed more diffusely as a result of the action of anthrax
toxin. 43-45 Microscopical examination of affected tissues reveals massive
edema and mucosal necrosis at infected sites. 45 Inflammatory infiltrates
are seen that are similar to those in cutaneous anthrax. Gram's staining of
peritoneal fluid may reveal numerous large gram-positive bacilli. 40,46 Although mediastinal
widening is considered pathognomonic of inhalational anthrax, it has also been
reported in a case of gastrointestinal anthrax. 47 Associated symptoms
include fever and diffuse abdominal pain with rebound tenderness. There are
reports of both constipation and diarrhea; the stools are either melenic or
blood-tinged. 46,48 Because of ulceration of the gastrointestinal mucosa, patients
often vomit material that is blood-tinged or has a coffeeground appearance.
Ascites develops with concomitant reduction in abdominal pain two to four days
after the onset of symptoms. The appearance of the ascites fluid ranges from
clear to purulent, and it often yields colonies of B. anthracis when cultured. Morbidity is due to blood loss,
fluid and electrolyte imbalances, and subsequent shock. Death results from
intestinal perforation or anthrax toxemia. If the patient survives, most of the
symptoms subside in 10 to 14 days. 48 Oropharyngeal anthrax is less common than the gastrointestinal
form. It is also associated with the ingestion of contaminated meat. Initial
symptoms include cervical edema and local lymphadenopathy, which cause
dysphagia and respiratory difficulties. Lesions can be seen in the oropharynx
and usually have the appearance of pseudomembranous ulcerations. This form is
milder than the classic gastrointestinal disease and has a more favorable
prognosis. 34,48
Inhalational Anthrax
Inhalational anthrax is rare, usually occurring
after the inhalation of pathogenic endospores from contaminated animal hides or
products. Before the introduction of hygienic measures in the 1960s, including
vaccination, workers in goathair mills, for example, were regularly exposed to
high concentrations of viable anthrax spores. Nevertheless, for reasons that
are not understood, few cases of inhalational anthrax occurred among them. 49-51 When dispersed in the
atmosphere as an aerosol, anthrax spores can present a respiratory hazard even
far downwind from the point of release, as demonstrated by animal tests on
Gruin-ard Island in the United Kingdom, 52-55 and by an accidental release from a military biologic facility in
the city of Sverdlovsk in the former Soviet Union. 2,56-58 Inhalational anthrax is
usually fatal, even with aggressive antimicrobial therapy. It appears that only
about one fifth of those who contracted inhalational anthrax in Sverdlovsk
recovered. 2 Anthrax spores are about 1 to 2 µm in diameter, a size that is
optimal for inhalation and deposition in the alveolar spaces. 51,59-61 Although the lung is the
initial site of contact, inhalational anthrax is not considered a true
pneumonia. In most but not all cases, there is no infection in the lungs. 58,62 Rather, the endospores
are engulfed by alveolar macrophages and transported by them to the mediastinal
and peribronchial lymph nodes, with the spores germinating en route. Anthrax
bacilli multiply in the lymph nodes, causing hemorrhagic mediastinitis, and
spread throughout the body in the blood. 43,62 Data from the Sverdlovsk
outbreak indicate a modal incubation time of approximately 10 days for
inhalational anthrax. However, the onset of symptoms occurred up to six weeks
after the reported date of exposure. 2,57 Such long incubation times presumably reflect the ability of
viable anthrax spores to remain in the lungs for many days. 51,63,64 Longer incubation
periods may be associated with smaller inocula. The initial symptoms most often
reported are fever, nonproductive cough, myalgia, and malaise, resembling those
of a viral upper respiratory tract infection. Early in the course of the
disease, chest radiographs show a widened mediastinum, which is evidence of
hemorrhagic mediastinitis, and marked pleural effusions. After one to three
days, the disease takes a fulminant course with dyspnea, strident cough, and
chills, culminating in death. 34,59 In Sverdlovsk, the mean time between the onset of symptoms and
death was 3 days (range, 1 to 10). Although accompanying evidence of clinical
signs of pneumonia in these cases is lacking, some of the autopsies from the
Sverdlovsk outbreak showed a focus of necrotizing hemorrhagic pneumonitis,
possibly at the portal of infection. 58 Submucosal hemorrhages occurred in the trachea and bronchi, with
hemorrhage and necrosis of peribronchial lymph nodes. Hemorrhagic mediastinal
lymph nodes represent the primary lesion; however, gastrointestinal and
leptomeningeal lesions are the result of hematogenous spread. There may be wide
individual variation in susceptibility to inhalational anthrax, as suggested by
experimental studies in nonhuman primates and by the absence of persons younger
than 24 years among the 66 deaths reported in the Sverdlovsk outbreak. 2,51,57
Anthrax Meningitis
Involvement of the meninges by B. anthracis is a rare complication of
anthrax. 65 The most common portal of entry is the skin, from which the
organisms can spread to the central nervous system by hematogenous or lymphatic
routes. Anthrax meningitis also occurs in cases of pulmonary and gastrointestinal
anthrax. 58,66 Anthrax meningitis is almost always fatal, with death occurring
one to six days after the onset of illness, despite intensive antibiotic
therapy.In the few cases in which patients have survived, antibiotic therapy
was combined with the administration of antitoxin, prednisone, or both. 65,67 In addition to common
meningeal symptoms and nuchal rigidity, the patient has fever, fatigue,
myalgia, headache, nausea, vomiting, and sometimes agitation, seizures, and
delirium. The initial signs are followed by rapid neurologic degeneration and
death. The pathological findings are consistent with a hemorrhagic meningitis,
with extensive edema, inflammatory infiltrates, and numerous grampositive
bacilli in the leptomeninges. 43,68 The cerebrospinal fluid is often bloody and contains many
gram-positive bacilli. 69 Gross examination at autopsy finds extensive hemorrhage of
theleptomeninges, which gives them a dark red appearance described as
cardinal's cap. 58
|
TABLE
1 |
DIFFERENTIAL
DIAGNOSIS OF CLINICAL MANIFESTATIONS OF ANTHRAX |
|
|
MANIFESTATION |
DISEASE |
CAUSATIVE ORGANISM |
|
Cutaneous
Anthrax |
Ecthyma gangrenosum |
Pseudomonas aeruginosa |
|
|
Rat-bite fever |
Streptobacillus
moniliformis, |
|
|
|
|
|
|
Ulceroglandular tularemia |
Francisella tularensis |
|
|
Plague |
Yersinia pestis |
|
|
Glanders |
Pseudomonas pseudomallei |
|
|
Rickettsialpox |
Rickettsia akari |
|
|
Orf |
Parapoxvirus |
|
|
Staphylococcal lymphadenitis |
Staphylococcus aureus |
|
|
Cutaneous tuberculosis |
Myocbacterium tuberculosis |
|
|
Leprosy |
Mycobacterium leprae |
|
|
Buruli ulcer |
Mycobacterium ulcerans |
|
Gastrointestinal
Anthrax |
Typhoid |
Salmonella typhi |
|
|
Intestinal tularemia |
Francisella tularensis |
|
|
Acute gastroenteritis |
|
|
|
Peritonitis |
|
|
|
Mechanical obstruction |
|
|
|
Peptic or duodenal ulcer |
|
|
Inhalational
Anthrax |
Acute bacterial mediastinitis |
|
|
|
Mycoplasmal pneumonia |
Mycoplasma pneumoniae |
|
|
Legionnaires disease |
Legionella pneumophila |
|
|
Psittacosis |
Chlamydia psittaci |
|
|
Tularemia |
Francisella tularensis |
|
|
Q fever |
Coxiella burnetii |
|
|
Viral pneumonia |
Influenzavirus, hantavirus, adenovirus, |
|
|
Histoplasmosis (fibrous mediastinitis) |
Histoplasma capsulatum |
|
|
|
|
|
|
Coccidioidomycosis |
Coccidioides immitis |
|
|
Ruptured aortic aneurysm |
|
|
|
Superior vena cava syndrome |
|
|
|
Silicosis |
|
|
|
Sarcoidosis |
|
|
Meningeal
Anthrax |
Subarachnoid hemorrhage |
|
DIAGNOSIS
Differential Diagnosis
Table 1 summarizes the differential diagnosis of
anthrax. In cutaneous anthrax, the painless, blackened, necrotic eschar is
limited to the late stages of the infection. The ulcerative eschar of cutaneous
anthrax must be differentiated from other papular lesions that present with
regional lymphadenopathy. If the lesion is purulent and the regional lymph
nodes are palpable, staphylococcal lymphadenitis is the most likely cause,
although cutaneous anthrax lesions can be superinfected with pyogenic bacteria.
70 The
initial symptoms of inhalational anthrax are nondescript or flulike and are
similar to those of atypical pneumonia from other causes. The prognosis is
improved if early treatment is implemented, so that a high level of suspicion
is necessary if there is a chance of exposure to anthrax. The cardiopulmonary
collapse associated with a history of radiographic evidence of mediastinal
widening in the late stages of inhalational anthrax must be differentiated from
cardiovascular collapse with noninfectious causes, such as dissecting or
ruptured aortic aneurysm and the superior vena cava syndrome. Anthrax infection
is unusual in that mediastinal changes can be detected early in the course of
infection by chest radiography, although similar pictures can be seen in acute
bacterial mediastinitis and fibrous mediastinitis due to Histoplasma capsulatum. 71 Less specific findings
include pleural effusions and radiographic evidence of pulmonary edema.
Silicosis, siderosis, alveolar proteinosis, and sarcoidosis are often
alternative causes of chronic mediastinitis in patients with the relevant
occupational history and previous chest radiographs demonstrating long-standing
mediastinal widening. When ingestion of contaminated meat is suspected, the symptoms
of an acute abdomen should be considered as possible early signs of intestinal
anthrax infection. Hemorrhagic meningitis caused by anthrax must be
distinguished from subarachnoid hemorrhage by computed tomography without
contrast. To distinguish hemorrhagic meningitis caused by B. anthracis from that caused by other
bacteria, Grams staining and culture of cerebrospinal fluid should be
performed. 68 In addition to the above indictors, the clinician should consider
anthrax if there is a history of contact with materials that may be
contaminated with spores, such as infected farm animals and imported hides, or
of travel to places where anthrax is endemic. Because of the remote possibility
of an anthrax aerosol attack, clinicians should be alert to any sudden deaths
of previously healthy persons from undiagnosed disease and report them promptly
to the Centers for Disease Control and Prevention and other appropriate public
health officials.
Bacteriologic Tests
B. anthracis is a nonmotile, gram-positive, aerobic rod 1.2 to 10 µm in length
and 0.5 to 2.5 µm in width that is capable of forming central or terminal
spores. It is part of the B. cereus group
of bacilli, which consists of B. cereus, B.
anthracis, B. thuringiensis, and B.
mycoides. 73 The bacteria in this group tend to be dismissed by clinical
microbiology laboratories as contaminants unless the physician specifically
requests testing. 73 Except for B. anthracis, all
members of this group are resistant to penicillin because they produce
chromosomally encoded betalactamases. 74 B. anthracis is easy to differentiate from other members of the B. cereus group by observing the
morphologic features of the colony on a blood-agar plate. Colonies of most B. anthracis isolates are nonhemolytic and
are white to gray, often looking like ground glass. 75 The unusually tenacious
colonies are able to retain their shape when manipulated. When inoculated onto
nutrient agar containing 0.7 percent bicarbonate and grown overnight at 37°C in
the presence of 5 to 20 percent carbon dioxide, B. anthracis will form its characteristic poly-D-glutamic acid capsule. 76 These colonies have a
mucoid appearance, and the capsule can be demonstrated microscopically in a
colony smear stained with McFadyean's polychrome methylene blue or India ink. 75 Blood samples obtained
from patients late in the course of infection and stained in the same manner
will reveal large numbers of encapsulated bacilli. Bacilli can also be observed
in and cultured from ascites fluid, pleural effusions, cerebrospinal fluid (in
cases of meningitis), 77 and fluid carefully expressed from the eschar, although expressing
eschar fluid is not recommended because it can cause dissemination of the
pathogen. 78 Patients with systemic disease often die before positive blood
cultures can be obtained, making early diagnosis and treatment crucial. If the
samples are likely to be contaminated with other bacillus species,
polymyxin-lysozyme-EDTA-thallous acetate agar is used as a selective medium for
B. anthracis. 79 The API 50 CH test strip
(API Laboratory Products, Plainview, N.Y.) can be used in conjunction with the
API 20E test strip to identify a number of bacillus species, including B. anthracis. 80 Blood cultures in cases
of systemic anthrax infection are almost always positive, because of the large
numbers of bacterial cells in the circulation. 1 Cultures of tissue from
skin lesions, however, are not useful diagnostically, because the rate of
positive cultures does not exceed 60 to 65 percent, probably owing to the use of
antimicro-bial therapy or the microbicidal activity of local antagonistic skin
flora. 81 There are reports of clinical isolates of B. anthracis that are resistant to
penicillin. 31,82 Because of the potential for drug-resistant strains, including
deliberately modified strains, antibiotic-susceptibility testing should be
performed on all isolates.
Serologic and Immunologic Tests
The major immunogenic proteins of B. anthracis appear to be capsular
antigens and the exotoxin components. Specific enzyme-linked immunosorbent
assays (ELISAs) that show a quadrupling of the titer of antibodies against
these components are diagnostic of past infection or vaccination. The most
reliable indicators are the titers of antibody to protective antigen and to
capsular components. 73,83,84 In studies of the measurement of antibody titers by ELISA, the
sensitivity of possible indicators was as follows: 72 percent for protective
antigen, 95 to 100 percent for capsule antigens, 42 percent for lethal factor,
and 26 percent for edema factor. 85 Enzyme-linked immunoelectrotransfer blotting provided a higher
specificity when used in conjunction with ELISA-based testing. 85 Indirect
microhemagglutination gives results similar to those obtained with ELISA but
has certain drawbacks, including the short shelf life of antigen-sensitized
red-cell preparations, the limited reproducibility of the test, and longer
preparation times. 86 Immunologic detection of the exotoxins in blood during systemic
infection is possible with similar tests if antibodies to anthrax toxins are
available, but those tests are unreliable for diagnosis. Thus, although these
tests are of epidemiologic value, they have little diagnostic value in acute
illness. 83 During systemic infections, antibodies to toxin or capsular
components cannot be detected until late in the course of the disease, often
when it is too late to initiate treatment. 73 In treated infections,
no increase in the antitoxin antibody titer is seen. The anthraxin skin test,
consisting of subdermal injection of a commercially produced chemical extract
of an attenuated strain of B. anthracis,
is available for the diagnosis of acute and previous cases of anthrax. 81,87,88 In one study the skin
test diagnosed 82 percent of cases one to three days after the onset of
symptoms and 99 percent of cases by the end of the fourth week. 81 The skin test may be
suitable for both rapid diagnosis of acute cases and the retrospective analysis
of anthrax infections.
New Molecular Diagnostic Methods
New diagnostic techniques have focused on the
use of the polymerase chain reaction to amplify markers specific to B. anthracis or the B. cereus group. Two markers, vrrA 89 and Ba813, 90-92 have been the subject of
extensive study. Other methods using the polymerase chain reaction to amplify
specific virulence plasmid markers harbored by different anthrax strains may
soon become available. 56,93-96 These new rapid methods may become useful in the clinical setting,
where early diagnosis is crucial.
PREVENTION AND TREATMENT
Prophylaxis, Vaccination, and Decontamination
Prophylaxis for asymptomatic patients with
suspected exposure to anthrax spores can be achieved with a six week course of
doxycycline or ciprofloxacin. If the suspected dose of spores is high, a longer
course of antibiotics is warranted. Extended treatment is needed for total
pulmonary clearance of spores, which are not affected by the presence of
antibiotics. 63,97 The standard anthrax vaccine in the United States is approved by
the Food and Drug Administration and is routinely administered to persons at
risk for exposure to anthrax spores. The existing supplies are currently being
used to immunize all military personnel. Designated anthrax vaccine adsorbed
(AVA), it is an aluminum hydroxide-precipitated preparation of protective
antigen from attenuated, nonencapsulated B.
anthracis cultures of the Sterne strain. 98,99 Two inoculations with
AVA afforded substantial protection against inhalational anthrax in rhesus
monkeys, 100 and a limited trial of a similar vaccine in humans indicated that
it afforded considerable protection against cutaneous anthrax. 101 AVA is administered
subcutaneously in a 0.5-ml dose that is repeated at 2 and 4 weeks and at 6, 12,
and 18 months. 102 Boosters are then given annually. For those receiving antibiotic
prophylaxis for suspected exposure, AVA may be given concurrently. There is a
need for vaccines with better protection and a simpler schedule. Vaccines now
being tested include preparations of protective antigen subunits with different
adjuvants, protective antigen purified from recombinant sources, and live
vaccines based on anthrax strains with auxotrophic mutations. 103-113 Live attenuated
endospore-based vaccines were widely used in the Soviet Union for both humans
and livestock and remain in use in the Russian Federation today. 103 The ability of any
vaccine to protect humans in the event of aerosol attack, as in biologic
terrorism or warfare, cannot be tested directly and therefore must remain a
concern. 114 A textile mill contaminated with anthrax spores was decontaminated
with vaporized formaldehyde, 115 and soil decontamination at Gruinard Island was achieved with
formaldehyde in seawater. 116 Although decontamination is desirable, the risk that resuspension
of a deposited aerosol will lead to inhalational anthrax is much less than the
risk due to a primary aerosol. 117,118 Autoclaving and incineration are acceptable procedures for the
decontamination of laboratory materials.
|
TABLE
2 |
Pharmacologic
Therapy for Bacillus anthracis Infection and Its Sequelae* |
|
|
Therapy |
Dosage
for Adults |
Dosage
for Children |
|
Treatment
of Infection† |
|
|
|
Penicillin
V |
200-500 mg orally 4 times/day |
25-50 mg/kg of body
weight/day orally in divided doses 2 or 4 times/day |
|
Penicillin
G |
8 million-12 million U total, intravenously in
divided doses every 4-6 hr, |
100,000-150,000 U/kg/day in
divided doses every 4-6 hr |
|
Streptomycin |
30 mg/kg intramuscularly or intravenously per
day - gentamicin can also be used (in conjunction with penicillin) |
|
|
Tetracycline |
250-500 mg orally or intravenously 4 times/day |
Tetracycline is not approved
for children |
|
Doxycycline |
200 mg orally or intravenously as a loading
dose, then 50-100 mg every 12 hr |
Doxycycline is not approved
for children <9 yr old For children Ç45 kg: 2.5 mg/kg every 12 hr For
children 45 kg: use adult dosage |
|
Erythromycin |
250 mg orally every 6 hr |
40/mg/kg/day orally in
divided doses every 6 hr |
|
Erythromycin
lactobionate |
15-20 mg/kg (maximum, 4 g) intravenously per
day |
20-40 mg/kg/day
intravenously in divided doses every 6 hr (1- to 2-hr infusion) |
|
Chloramphenicol |
50-100 mg/kg/day orally or intravenously in
divided doses every 6 hr |
50-75 mg/kg/day in divided
doses every 6 hr |
|
Ciprofloxacin |
250-750 mg orally twice/day 200-400 mg
intravenously every 12 hr |
20-30 mg/kg/day in divided
doses every 12 hr Oral or intravenous dosing is not approved for patients
<18 yr old |
|
Prophylaxis‡ |
|
|
|
Doxycycline |
100 mg orally twice/day for 4 wk |
|
|
Ciprofloxacin |
500 mg orally twice/day for 4 wk |
|
|
Corticosteroid therapy for severe edema |
|
|
|
Dexamethasone |
0.75-0.90 mg/kg/day |
0.25-0.5 mg/kg every 6 hr |
|
Prednisone |
1-2 mg/kg or 5-60 mg orally/day |
0.5-2 mg/kg/day |
|
*Most B. anthracis strains are resistant to
cefuroxime in vitro. †For inhalational, gastrointestinal, or
meningeal anthrax infection in adults, the intravenous regimen is used with
peni- cillin G, streptomycin, tetracycline, doxycycline, erythromycin lactobionate,
chloramphenicol, and ciprofloxacin; for these infections in children, the
intravenous regimen is used with penicillin G, doxycycline, erythromycin
lactobionate, and chloramphenicol. ‡If patient is unvaccinated, begin initial
doses of vaccine. |
||
Treatment
Antibiotics
Table 2 summarizes pharmacologic therapy for
anthrax. Penicillin and doxycycline are used for the treatment of anthrax.
Intravenous administration is recommended in cases of inhalational,
gastrointestinal, and meningeal anthrax. Cutaneous anthrax with signs of
systemic involvement, extensive edema, or lesions on the head and neck also
requires intravenous therapy. Streptomycin had a synergistic effect with
penicillin in experiments and may also be given for inhalational anthrax.
Despite early and vigorous treatment, the prognosis of patients with
inhalational, gastrointestinal, or meningeal anthrax remains poor. Antibiotic
therapy should be continued for at least 14 days after symptoms abate. 67,78 In cutaneous anthrax,
treatment with oral penicillin renders lesions sterile after 24 hours, although
they still progress to eschar formation. Chloramphenicol, erythromycin,
tetracycline, or ciprofloxacin can be administered to patients who are allergic
to penicillin. If resistance to penicillin and doxycycline is suspected and
antibiotic-susceptibility data are not available, ciprofloxacin may be
administered empirically. Doxycycline and tetracycline are not recommended for
pregnant women or children, and the effects of ciprofloxacin in pregnant women
have not been determined. 4 For culturing cutaneous lesions, gentle sampling with a moist,
sterile applicator is preferred. Excision of the eschar is contraindicated and
might hasten systemic dissemination. Lesions should be covered with sterile
dressings that are changed regularly. Soiled dressings should be autoclaved and
properly disposed of. In cases of extensive edema, meningitis, or swelling in
the head-and-neck region, corticosteroid therapy should be initiated. 119,120 Supportive therapy
should be initiated to prevent septic shock and fluid and electrolyte
imbalance, and to maintain airway patency.
Potential New Treatments
The current understanding that anthrax is a
toxigenic condition suggests the potential of antitoxin
therapy. The central importance of lethal toxin
is supported by much research. Early experiments in which antibiotics were
administered to animals at different stages of infection found a principle of
no return; once the infection had reached a certain point, the animal was
doomed, even after removal of the microbes. Test animals injected intravenously
with purified lethal toxin died in a manner very similar to that of animals
that died of the natural infection. 3,15,19 Lethal-toxin-deficient strains are highly attenuated. 121,122 Prior immunity (passive
or active) to the lethal-toxin proteins protects animals from endospore
challenge. 63,123 Finally, toxin-affected macrophages produce the proinflammatory
cytokines that mediate the shock and sudden death that occur in anthrax. 3,15,19 Unfortunately, antitoxin
preparations are not currently available in the United States. In addition, the
recent discovery that lethal toxin acts as a zinc metalloprotease inside target
cells and the identification of potential target substrates may provide new
insights for use in designing drugs that directly inhibit the toxicity of
lethal factor in vivo. 14,17,18824 á September 9, 1999
FUTURE CHALLENGES
Anthrax holds an important place in the
development of modern medicine and has long been intertwined with human
history. Anthrax is believed to have been one of the Egyptian plagues at the
time of Moses, and cases were clearly recorded by the ancient Romans. 124 The anthrax bacillus was
the model first used in the development of Koch's postulates and is considered
the first germ proved to cause human disease. 125 Pasteur later generated
a capsule-null anthrax strain that was the first vaccine made from live
attenuated bacteria for use in humans. 126 At the birth of cellular immunology, Metchnikoff used the anthrax
bacillus to examine the ability of his newly discovered macrophages to kill
microbes. 127 Today, investigators are using B.
anthracis and its toxins in an attempt to understand early events in
the infectious process and the molecular basis of inflammation. 3,15,19 Unfortunately, new
issues have arisen beyond those related to scientific inquiry. No
casualty-producing terrorist use of anthrax has occurred, and the Federal
Bureau of Investigation has stated that it has no intelligence that state
sponsors of terrorism, international terrorist groups, or domestic terrorist
groups are currently planning to use these deadly weapons in the United States.
128 However,
the incidence of hoaxes has greatly increased with recent publicity about
anthrax, providing a challenge to law enforcement. 129 Recent revelations
regarding the development of anthrax weapons by the former Soviet Union and by
Iraq, and of attempts to develop such weapons by the Aum Shinrikyo cult in
Japan, make the potential use of B.
anthracis in biologic terrorism a legitimate concern. 4,129 New strains resistant to
antibiotics or containing additional virulence factors could be misused with the
intent of confounding treatment or prophylaxis. 114,130 Whether our medical
system would be able to provide appropriate prophylaxis and therapy in the
event of a large-scale exposure to pathogenic endospores remains uncertain,
even doubtful. It has now become relevant for physicians to re-familiarize
themselves with clinical anthrax.
Supported in part by grants (AI-08649 and
AI-40644) and a Medical Scientist Training award from the National Institutes
of Health, by a grant (IRG-158 K) from the American Cancer Society, and by Duke
University Medical Center.
We are indebted to Arthur
Friedlander, M.D., Julia Chosy, Tanya Dixon, John Ireland, Matthew Weiner, and
Kenneth Alexander, M.D., Ph.D., for their reading and critical discussion of
the manuscript.
REFERENCES
1. Hanna P. Anthrax pathogenesis and host response. Curr Top
Microbiol Immunol 1998;225:13-35.
2. Meselson M, Guillemin J, Hugh-Jones M, et al. The Sverdlovsk
anthrax outbreak of 1979. Science 1994;266:1202-8.
3. Hanna PC, Acosta D, Collier RJ. On the role of macrophages in
anthrax. Proc Natl Acad Sci U S A 1993;90:10198-201.
4. Inglesby TV, Henderson DA, Bartlett JG, et al. Anthrax as a
biological weapon: medical and public health management. JAMA 1999;281:1735-45.
5. Watson A, Keir D. Information on which to base assessments of risk
from environments contaminated with anthrax spores. Epidemiol Infect
1994;113:479-90.
6. Harris SH. Factories of death: Japanese secret biological warfare,
1932-1945, and the American cover-up. London: Routledge, 1994.
7. Ross JM. The pathogenesis of anthrax following the administration
of spores by the respiratory route. J Pathol Bacteriol 1957;73:485-94.
8. Guidi-Rontani C, Weber-Levy M, Labruyere E, Mock M. Germination of
Bacillus anthracis spores within alveolar macrophages. Mol Microbiol
1999;31:9-17.
9. Leppla SH. The anthrax toxin complex. In: Alouf J, Freer JH,
eds.Sourcebook of bacterial protein toxins. London: Academic Press,
1991:277-302.
10. Idem. Anthrax toxin edema factor: a bacterial adenylate cyclase that
increases cyclic AMP concentrations of eukaryotic cells. Proc Natl Acad Sci U S
A 1982;79:3162-6.
11. Idem. Bacillus anthracis calmodulin-dependent adenylate cyclase:
chemical and enzymatic properties and interactions with eucaryotic cells. Adv
Cyclic Nucleotide Protein Phosphorylation Res 1984;17:189-98.
12. O'Brien J, Friedlander A, Dreier T, Ezzell J, Leppla S. Effects of
anthrax toxin components on human neutrophils. Infect Immun 1985;47:306-10.
13. Alexeyev OA, Morozov VG, Suzdaltseva TV, Mishukov AS, Steinberg
LA. Impaired neutrophil function in the cutaneous form of anthrax. Infection
1994;22:281-2.
14. Hammond SE, Hanna PC. Lethal factor active-site mutations affect
catalytic activity in vitro. Infect Immun 1998;66:2374-8.
15. Hanna PC, Kruskal BA, Ezekowitz RA, Bloom BR, Collier RJ. Role of
macrophage oxidative burst in the action of anthrax lethal toxin. Mol Med
1994;1:7-18.
16. Klimpel KR, Arora N, Leppla SH. Anthrax toxin lethal factor
contains a zinc metalloprotease consensus sequence which is required for lethal
toxin activity. Mol Microbiol 1994;13:1093-100.
17. Duesbery NS, Webb CP, Leppla SH, et al. Proteolytic inactivation
of MAP-kinase-kinase by anthrax lethal factor. Science 1998;280:734-7.
18. Vitale G, Pellizzari R, Recchi C, Napolitani G, Mock M, Montecucco
C. Anthrax lethal factor cleaves the N-terminus of MAPKKs and induces
tyrosine/threonine phosphorylation of MAPKs in cultured macrophages. Biochem
Biophys Res Commun 1998;248:706-11.
19. Hanna PC, Kochi S, Collier RJ. Biochemical and physiological
changes induced by anthrax lethal toxin in J774 macrophage-like cells. Mol Biol
Cell 1992;3:1269-77.
20. Makino S-I, Uchida I, Terakado N, Sasakawa C, Yoshikawa M.
Molecular characterization and protein analysis of the cap region, which is essential for
encapsulation in Bacillus anthracis.
J Bacteriol 1989;171:722-30.
21. Makino S, Sasakawa C, Uchida I, Terakado N, Yoshikawa M. Cloning
and CO2 -dependent
expression of the genetic region for encapsulation from Bacillus anthracis. Mol Microbiol
1988;2:371-6.
22. Dai Z, Sirard J-C, Mock M, Koehler TM. The atxA gene product activates transcription
of the anthrax toxin genes and is essential for virulence. Mol Microbiol
1995;16:1171-81.
23. Dai Z, Koehler TM. Regulation of anthrax toxin activator gene
(atxA) expression in Bacillus anthracis: temperature, not CO2/bicarbonate,
affects AtxA synthesis. Infect Immun 1997;65:2576-82.
24. Uchida I, Hornung JM, Thorne CB, Klimpel KR, Leppla SH. Cloning
and characterization of a gene whose product is a transactivator of anthrax
toxin synthesis. J Bacteriol 1993;175:5329-38.
25. Uchida I, Makino S, Sekizaki T, Terakado N. Cross-talk to the
genes for Bacillus anthracis capsule synthesis by atxA, the gene encoding the
transactivator of anthrax toxin synthesis. Mol Microbiol 1997;23:1229-40.
26. Vietri NJ, Marrero R, Hoover TA, Welkos SL. Identification and
characterization of a transactivator involved in the regulation of
encapsulation by Bacillus anthracis. Gene
1995;152:1-9.
27. Taylor JP, Dimmitt DC, Ezzell JW, Whitford H. Indigenous human
cutaneous anthrax in Texas. South Med J 1993;86:1-4.
28. Human cutaneous anthrax North Carolina, 1987. Arch Dermatol
1988;124:1324.
29. Leads from the MMWR: human cutaneous anthrax -- North Carolina,
1987. JAMA 1988;260:616.
30. Human cutaneous anthrax -- North Carolina, 1987. MMWR Morb Mortal
Wkly Rep 1988;37:413-4.
31. Bradaric N, Punda-Polic V. Cutaneous anthrax due to
penicillin-resistant Bacillus anthracis transmitted by an insect bite. Lancet
1992;340:306-7.
32. Turell MJ, Knudson GB. Mechanical transmission of Bacillus
anthracis by stable flies (Stomoxys calcitrans) and mosquitoes (Aedes aegypti
and Aedes taeniorhynchus). Infect Immun 1987;55:1859-61.
33. Smego RA Jr, Gebrian B, Desmangels G. Cutaneous manifestations of
anthrax in rural Haiti. Clin Infect Dis 1998;26:97-102.
34. Edwards MS. Anthrax. In: Feigin RD, Cherry JD, eds. Textbook of
pediatric infectious diseases. 3rd ed. Vol. 1. Philadelphia: W.B.
Saunders, 1992:1053-6.
35. Doganay M, Bakir M, Dokmetas I. A case of cutaneous anthrax with
toxaemic shock. Br J Dermatol 1987;117:659-62.
36. Kutluk MT, Secmeer G, Kanra G, Celiker A, Aksoyek H. Cutaneous
anthrax. Cutis 1987;40:117-8.
37. Doganay M, Aygen B, Inan M, Kandemir O, Turnbull P. Temporal
artery inflammation as a complication of anthrax. J Infect 1994;28:311-4.
38. Yorston D, Foster A. Cutaneous anthrax leading to corneal scarring
from cicatricial ectropion. Br J Ophthalmol 1989;73:809-11.
39. Mallon E, McKee PH. Extraordinary case report: cutaneous
anthrax.Am J Dermatopathol 1997;19:79-82.
40. LaForce FM. Anthrax. Clin Infect Dis 1994;19:1009-14.
41. de Lalla F, Ezzell JW, Pellizzer G, et al. Familial outbreak of
agriculture anthrax in an area of northern Italy. Eur J Clin Microbiol Infect
Dis 1992;11:839-42.
42. Batykin V, Vygodchikov G, Sazshina Y. Outbreak of intestinal
anthrax in Yarolslavl. J Hyg Epidemiol 1929;1:25-30.
43. Dutz W, Kohout E. Anthrax. Pathol Annu 1971;6:209-48.
44. Dutz W, Saidi F, Kohout E. Gastric anthrax with massive ascites.
Gut 1970;11:352-4.
45. Dutz W, Kohout-Dutz E. Anthrax. Int J Dermatol 1981;20:203-6.
46. Nalin DR, Sultana B, Sahunja R, et al. Survival of a patient with
intestinal anthrax. Am J Med 1977;62:130-2.
47. Paulet R, Caussin C, Coudray JM, Selcer D, de Rohan Chabot P.
Forme viscerale de charbon humain importee d'Africa. Presse Med 1994; 23:477-8.
48. Alizad A, Ayoub EM, Makki N. Intestinal anthrax in a two-year-old
child. Pediatr Infect Dis J 1995;14:394-5.
49. Dahlgren CM, Buchanan LM, Decker HM, Freed SW, Phillips CR,
Brachman PS. Bacillus anthracis aerosols
in goat hair processing mills. Am J Hyg 1960;72:24-31.
50. Albrink WS, Brooks SM, Biron RE, Kopel M. Human inhalation
anthrax: a report of three fatal cases. Am J Pathol 1960;36:457-71.
51. Brachman PS, Kaufman AF, Dalldorf FG. Industrial inhalation
anthrax. Bacteriol Rev 1966;30:646-59.
52. Manchee RJ, Broster MG, Henstridge RM, Stagg AJ, Melling J.
Anthrax island. Nature 1982;296:598.
53. Manchee RJ. Bacillus anthracis on
Gruinard Island. Nature 1981;294: 254-5.
54. Sterne M. Anthrax island. Nature 1982;295:362.
55. Wynn J. Anthrax island: why worry? Nature 1982;298:506-7.
56. Jackson PJ, Hugh-Jones ME, Adair DM, et al. PCR analysis of tissue
samples from the 1979 Sverdlovsk anthrax victims: the presence of multiple
Bacillus anthracis strains in different victims. Proc Natl Acad Sci U S A
1998;95:1224-9.
57. Guillemin J. Anthrax: the investigation of a deadly outbreak.
Berkeley: University of California Press (in press).
58. Abramova FA, Grinberg LM, Yampolskaya OV, Walker DH. Pathology of
inhalational anthrax in 42 cases from the Sverdlovsk outbreak of 1979. Proc
Natl Acad Sci U S A 1993;90:2291-4.
59. Penn CC, Klotz SA. Anthrax pneumonia. Semin Respir Infect 1997;
12:28-30.
60. Brachman PS. Inhalation anthrax. Ann N Y Acad Sci 1980;353:83-93.
61. Idem. Anthrax. Ann N Y Acad Sci 1970;174:577-82.
62. Albrink WS. Pathogenesis of inhalation anthrax. Bacteriol Rev
1961; 25:268-73.
63. Friedlander AM, Welkos SL, Pitt ML, et al. Postexposure
prophylaxis against experimental inhalation anthrax. J Infect Dis
1993;167:1239-43.
64. Henderson DW, Peacock S, Belton FC. Observations on the prophylaxis
of experimental pulmonary anthrax in the monkey. J Hyg 1956;54: 28-36.
65. Tahernia AC, Hashemi G. Survival in anthrax meningitis. Pediatrics
1972;50:329-33.
66. Tabatabaie P, Syadati A. Bacillus anthracis as a cause of
bacterial meningitis. Pediatr Infect Dis J 1993;12:1035-7.
67. Tahernia AC. Treatment of anthrax in children. Arch Dis Child
1967; 42:181-2.
68. Rangel RA, Gonzalez DA. Bacillus anthracic meningitis. Neurology
1975;25:525-30.
69. Pluot M, Vital C, Aubertin J, Croix JC, Pire JC, Poisot D. Anthrax
meningitis: report of two cases with autopsies. Acta Neuropathol (Berl)
1976;36:339-45.
70. Aksaray N, Cinaz P, Coskun U, Serbest M, Koksal F. Cutaneous
anthrax. Trop Geogr Med 1990;42:168-71.
71. Wheat J. Histoplasma. In: Gorbach SL, Bartlett JG, Blacklow NR,
eds.Infectious diseases. 2nd ed. Philadelphia: W.B. Saunders,
1998:2335-44.
72. Claus D, Berkeley RCW. Genus Bacillus.
In: Sneath PHA, ed. Bergey's manual of systematic bacteriology. Vol.
2. Baltimore: Williams & Wilkins, 1986:1105-39.
73. Turnbull PCB, Kramer JM. Bacillus.
In: Balows A, ed. Manual of clinical microbiology. 5th
ed. Washington, D.C.: American Society for Microbiology, 1991:296-303.
74. Penn CC, Klotz SA. Bacillus
anthracis and other aerobic spore formers. In: Gorbach SL, Bartlett
JG, Blacklow NR, eds. Infectious diseases. 2nd ed. Philadelphia:
W.B. Saunders, 1998:1747-50.
75. Parry JM, Turnbull PCB, Gibson JR. A color atlas of Bacillus species. London: Wolfe Medical,
1983:272.
76. Green BD, Battisti L, Koehler TM, Thorne CB, Ivins BE.
Demonstration of a capsule plasmid in Bacillus
anthracis. Infect Immun 1985;49:291-7.
77. Penn CC, Klotz SA. Anthrax. In: Gorbach SL, Bartlett JG, Blacklow
NR, eds. Infectious diseases. 2nd ed. Philadelphia: W.B. Saunders,
1998: 1575-8.
78. Gold H. Treatment of anthrax. Fed Proc 1967;26:1563-8.
79. Knisely RF. Selective medium for Bacillus
anthracis. J Bacteriol 1966; 92:784-6.
80. Logan NA, Carman JA, Melling J, Berkeley RCW. Identification of Bacillus anthracis by API tests. J Med
Microbiol 1985;20:75-85.
81. Shlyakhov E, Rubinstein E. Evaluation of the anthraxin skin test
for diagnosis of acute and past human anthrax. Eur J Clin Microbiol Infect Dis
1996;15:242-5.
82. Lalitha MK, Thomas MK. Penicillin resistance in Bacillus
anthracis. Lancet 1997;349:1522.
83. Harrison LH, Ezzell JW, Abshire TG, Kidd S, Kaufmann AF.
Evaluation of serologic tests for diagnosis of anthrax after an outbreak of
cutaneous anthrax in Paraguay. J Infect Dis 1989;160:706-10.
84. Turnbull PC, Doganay M, Lindeque PM, Aygen B, McLaughlin J.
Serology and anthrax in humans, livestock and Etosha National Park wildlife.
Epidemiol Infect 1992;108:299-313.
85. Sirisanthana T, Nelson KE, Ezzell JW, Abshire TG. Serological
studies of patients with cutaneous and oraloropharyngeal anthrax from northern
Thailand. Am J Trop Med Hyg 1988;39:575-81.
86. Johnson-Winegar A. Comparison of enzyme-linked immunosorbent and
indirect hemagglutination assays for determining anthrax antibodies. J Clin
Microbiol 1984;20:357-61.
87. Pfisterer RM. Eine Milzbrandepidemie in der Schweiz: Klinische,
diagnostische und epidemiologische Aspekte einer weitgehend vergessenen
Krankheit. Schweiz Med Wochenschr 1991;121:813-25.
88. Shlyakhov E, Rubinstein E, Novikov I. Anthrax post-vaccinal
cell-mediated immunity in humans: kinetics pattern. Vaccine 1997;15:631-6.
89. Andersen GL, Simchock JM, Wilson KH. Identification of a region of
genetic variability among Bacillus anthracis
strains and related species. J Bacteriol 1996;178:377-84.
90. Patra G, Vaissaire J, Weber-Levy M, Le Doujet C, Mock M. Molecular
characterization of Bacillus strains
involved in outbreaks of anthrax in France in 1997. J Clin Microbiol
1998;36:3412-4.
91. Patra G, Sylvestre P, Ramisse V, Therasse J, Guesdon JL. Isolation
of a specific chromosomic DNA sequence of Bacillus anthracis and its possible
use in diagnosis. FEMS Immunol Med Microbiol 1996;15:223-31.
92. Ramisse V, Patra G, Garrigue H, Guesdon JL, Mock M. Identification
and characterization of Bacillus anthracis by multiplex PCR analysis of
sequences on plasmids pXO1 and pXO2 and chromosomal DNA. FEMS Microbiol Lett
1996;145:9-16.
93. Keim P, Kalif A, Schupp J, et al. Molecular evolution and
diversity in Bacillus anthracis as detected by amplified fragment length
polymorphism markers. J Bacteriol 1997;179:818-24.
94. Klevytskya A, Schupp J, Price LB, et al. A multiplexed fluorescent
PCR genotyping system based on ten highly informative single-locus markers for B. anthracis strain identification.
Presented at the Third International Conference on Anthrax, Plymouth, England,
1998. abstract.
95. Price LB, Hugh-Jones M, Jackson PJ, Keim P. Genetic diversity in
the protective antigen gene of Bacillus anthracis. J Bacteriol
1999;181:2358-62.
96. Jackson PJ, Walthers EA, Kalif AS, et al. Characterization of the
variable-number tandem repeats in vrrA from different Bacillus anthracis
isolates. Appl Environ Microbiol 1997;63:1400-5.
97. Vancurik J. Causes of the failure of antibiotic prophylaxis of
inhalation anthrax and clearance of the spores from the lungs. Folia Microbiol
(Praha) 1966;11:459-64.
98. Vaccine against anthrax. BMJ 1965;5464:717-8.
99. Anthrax vaccine. Med Lett Drugs Ther 1998;40:52-3.
100. Ivins BE, Fellows P, Pitt ML, et al. Efficacy of standard human
anthrax vaccine against Bacillus anthracis aerosol
spore challenge in rhesus monkeys. Salisbury Med Bull 1996;87:125-6.
101. Brachman PS, Gold H, Plotkin SA, Fekety FR, Werrin M, Ingraham NR.
Field evaluation of a human anthrax vaccine. Am J Public Health 1962;52:632-45.
102. Franz DR, Jahrling PB, Friedlander AM, et al. Clinical recognition
and management of patients exposed to biological warfare agents. JAMA
1997;278:399-411.
103. Shlyakhov EN, Rubinstein E. Human live anthrax vaccine in the
former USSR. Vaccine 1994;12:727-30.
104. Pezard C, Weber M, Sirard JC, Berche P, Mock M. Protective
immunity induced by Bacillus anthracis toxin-deficient strains. Infect Immun
1995;63:1369-72.
105. Pezard C, Sirard JC, Mock M. Protective immunity induced by
Bacillus anthracis toxin mutant strains. Adv Exp Med Biol 1996;397:69-72.
106. Miller J, McBride BW, Manchee RJ, Moore P, Baillie LW. Production
and purification of recombinant protective antigen and protective efficacy
against Bacillus anthracis. Lett Appl Microbiol 1998;26:56-60.
107. Little SF, Knudson GB. Comparative efficacy of Bacillus anthracis
live spore vaccine and protective antigen vaccine against anthrax in the guinea
pig. Infect Immun 1986;52:509-12.
108. Ivins BE, Welkos SL, Knudson GB, Little SF. Immunization against
anthrax with aromatic compound-dependent (Aro-) mutants of Bacillus anthracis
and with recombinant strains of Bacillus subtilis that produce anthrax
protective antigen. Infect Immun 1990;58:303-8.
109. Ivins BE, Welkos SL, Little SF, Crumrine MH, Nelson GO.
Immunization against anthrax with Bacillus anthracis protective antigen
combined with adjuvants. Infect Immun 1992;60:662-8.
110. Ivins BE, Fellows PF, Nelson GO. Efficacy of a standard human
anthrax vaccine against Bacillus anthracis spore challenge in guinea-pigs.
Vaccine 1994;12:872-4.
111. Coulson NM, Fulop M, Titball RW. Bacillus anthracis protective
antigen, expressed in Salmonella typhimurium SL 3261, affords protection
against anthrax spore challenge. Vaccine 1994;12:1395-401.
112. Baillie L, Moir A, Manchee R. The expression of the protective
antigen of Bacillus anthracis in Bacillus subtilis. J Appl Microbiol 1998;84:
741-6.
113. Baillie LW, Moore P, McBride BW. A heat-inducible Bacillus
subtilis bacteriophage phi 105 expression system for the production of the
protective antigen of Bacillus anthracis. FEMS Microbiol Lett 1998;163:43-7.
114. Hanna P. How anthrax kills. Science 1998;280:1671, 1673-4.
115. Young LS, Feeley JC, Brachman PS. Vaporized formaldehyde treatment
of a textile mill contaminated with Bacillus anthracis. Arch Environ Health
1970;20:400-3.
116. Manchee RJ, Broster MG, Stagg AJ, Hibbs SE. Formaldehyde dilution
effectively inactivates spores of Bacillus
anthracis on the Scottish island of Gruinard. Appl Environ Microbiol
1994;60:4167-71.
117. Birensvige A. Inhalation hazard from reaerosolized biological
agents: a review. Aberdeen, Md.: Army Chemical Research, Development and
Engineering Center, 1992.
118. Davids DE, Lejeune AR. Secondary aerosol hazard in the field.
Raulston, Alta.: Suffield Defence Research Establishment, 1981.
119. Knudson GB. Treatment of anthrax in man: history and current
concepts. Mil Med 1986;151:71-7.
120. Doust JY, Sarkarzadeh A, Kavoossi K. Corticosteroid in treatment
of malignant edema of chest wall and neck (anthrax). Dis Chest 1968;53:773-4.
121. Cataldi A, Labruyere E, Mock M. Construction and characterization
of a protective antigen-deficient Bacillus anthracis strain. Mol Microbiol
1990;4:1111-7.
122. Pezard C, Berche P, Mock M. Contribution of individual toxin
components to virulence of Bacillus anthracis. Infect Immun 1991;59:3472-7.
123. Turnbull PC, Leppla SH, Broster MG, Quinn CP, Melling J.
Antibodies to anthrax toxin in humans and guinea pigs and their relevance to
protective immunity. Med Microbiol Immunol (Berl) 1988;177:293-303.
124. Dirckx JH. Virgil on anthrax. Am J Dermatopathol 1981;3:191-5.
125. Koch R. The aetiology of anthrax based on the ontogeny of the
an-thrax bacillus. Beitr Biol Pflanz 1877;2:277-82.
126. Pasteur L. De l'attenuation des virus et de leur retour ˆ la
virulence. C R Acad Sci III 1881;92:429-35.
127. Metchnikoff E. Immunity in infective diseases. Cambridge, England:
Cambridge University Press, 1905.
128. Ember LR. Bioterrorism: combating the threat. Chem Eng News
1999;77:8-17.
129. Bioterrorism alleging use of anthrax and interim guidelines for
management -- United States, 1998. MMWR Morb Mortal Wkly Rep 1999; 48:69-74.
130. Pomerantsev AP, Staritsin NA, Mockov Yu V, Marinin LI. Expression
of cereolysine AB genes in Bacillus anthracis vaccine strain ensures protection
against experimental hemolytic anthrax infection. Vaccine 1997;15: 1846-50.
Edward E.
Rylander, M.D.
Diplomat American
Board of Family Practice.
Diplomat American
Board of Palliative Medicine.