BMJ 2001;323:773 ( 6 October )
Papers
Early enteral feeding versus "nil by mouth" after
gastrointestinal surgery: systematic review and meta-analysis of controlled
trials
Stephen J Lewis
a Department of Medicine,
Addenbrooke's Hospital, Cambridge CB2 2QQ, b MRC Health
Services Research Collaboration, Department of Social Medicine, University of
Bristol, Bristol BS8 2PR, c Department of Surgery, Bristol
Royal Infirmary, Bristol BS2 8HW, d Department of Maxillofacial
Surgery, University of Bristol, Bristol BS1 2LY
Correspondence to: S Lewis [log in to unmask]
|
|
Abstract |
Objective: To determine whether a period of
starvation (nil by mouth) after gastrointestinal surgery is
beneficial in terms of specific outcomes.
Design: Systematic review and
meta-analysis of randomised controlled trials comparing any type of
enteral feeding started within 24 hours after surgery with nil
by mouth management in elective gastrointestinal surgery. Three
electronic databases (PubMed, Embase, and the Cochrane controlled
trials register) were searched, reference lists checked, and letters
requesting details of unpublished trials and data sent to
pharmaceutical companies and authors of previous trials.
Main outcome measures: Anastomotic
dehiscence, infection of any type, wound infection, pneumonia,
intra-abdominal abscess, length of hospital stay, and mortality.
Results: Eleven studies with
837 patients met the inclusion criteria. In six studies
patients in the intervention group were fed directly into the small
bowel and in five studies patients were fed orally. Early feeding
reduced the risk of any type of infection (relative risk
0.72, 95% confidence interval 0.54 to 0.98, P=0.036)
and the mean length of stay in hospital (number of days reduced by
0.84, 0.36 to 1.33, P=0.001). Risk reductions were
also seen for anastomotic dehiscence (0.53, 0.26 to 1.08, P=0.080),
wound infection, pneumonia, intra-abdominal abscess, and mortality,
but these failed to reach significance (P>0.10). The risk of
vomiting was increased among patients fed early (1.27, 1.01 to
1.61, P=0.046).
Conclusions: There seems to be no
clear advantage to keeping patients nil by mouth after elective
gastrointestinal resection. Early feeding may be of benefit. An
adequately powered trial is required to confirm or refute the
benefits seen in small trials.
|
What is already known on this topic Theoretically, early enteral feeding
improves tissue healing and reduces septic complications after
gastrointestinal surgery What this study adds Septic complications and length of
hospital stay were reduced in those patients who received early enteral
feeding In patients who received early enteral feeding
there were no significant reductions in incidence of anastomotic dehiscence,
wound infection, pneumonia, intra-abdominal abscess, and mortality |
|
|
Introduction |
A period of starvation ("nil by mouth") is common
practice after gastrointestinal surgery during which an intestinal anastomosis
has been formed. The stomach is decompressed with a nasogastric tube
and intravenous fluids are given, with oral feeding being introduced
as gastric dysmotility resolves.1 The
rationale of nil by mouth is to prevent postoperative nausea and
vomiting and to protect the anastomosis, allowing it time to heal
before being stressed by food. It is, however, unclear whether
deferral of enteral feeding is beneficial.
Contrary to widespread opinion, evidence from clinical studies and
animal experiments suggests that initiating feeding early is
advantageous. Postoperative dysmotility predominantly affects the
stomach and colon, with the small bowel recovering normal function
4-8 hours after laparotomy.1 Feeding
within 24 hours after laparotomy is tolerated and the feed
absorbed. 2
3
Gastrointestinal surgery is often undertaken in patients who are
malnourished,4-6
which in severe cases is known to increase morbidity.7 In
animals, starvation reduces the collagen content in anastomotic scar
tissue 8 9 and
diminishes the quality of healing, 9 10 whereas
feeding reverses mucosal atrophy induced by starvation11 and
increases anastomotic collagen deposition and strength.12
Experimental data in both animals and humans suggest that enteral
nutrition is associated with an improvement in wound healing.13 Finally,
early enteral feeding may reduce septic morbidity after abdominal trauma14 and
pancreatitis.15
Several clinical trials directly comparing strategies of early
feeding with nil by mouth after elective gastrointestinal surgery have
been performed. These studies, however, have not been systematically reviewed.
We performed a systematic review and meta-analysis of randomised
trials to assess the evidence on benefit and harm of early enteral
feeding.
|
|
Methods |
Eligibility criteria and literature
search![]() Clinical trials
were eligible if patients had undergone elective gastrointestinal surgery
and were randomly allocated to receive either enteral feeding
(within 24 hours after surgery) or the traditional management of
nil by mouth and intravenous fluids with introduction of enteral fluids
and diet as tolerated. We supplemented computerised searches of
PubMed, Embase, and the Cochrane controlled trials register with
checks of relevant reference lists. We wrote to trialists requesting
additional data on outcomes not reported in publications and on
trial methods. We also approached pharmaceutical companies that
produce enteral feeds and asked whether they held data from unpublished
trials.
Clinical trials
were eligible if patients had undergone elective gastrointestinal surgery
and were randomly allocated to receive either enteral feeding
(within 24 hours after surgery) or the traditional management of
nil by mouth and intravenous fluids with introduction of enteral fluids
and diet as tolerated. We supplemented computerised searches of
PubMed, Embase, and the Cochrane controlled trials register with
checks of relevant reference lists. We wrote to trialists requesting
additional data on outcomes not reported in publications and on
trial methods. We also approached pharmaceutical companies that
produce enteral feeds and asked whether they held data from unpublished
trials.
Data extraction and outcomes![]() From each study we collected data on the site of surgery, whether
an intestinal anastomosis was formed, whether the pathology was
benign or malignant, the type of feed used, and the method of
administration of the feed. The site of surgery was classified as
pancreatic, hepatobiliary, upper gastrointestinal (proximal to the
jejunum), or lower gastrointestinal (distal to the duodenum).
Outcomes potentially related to feeding included anastomotic
dehiscence, infection of any type, wound infection, pneumonia,
intra-abdominal abscess, vomiting, mortality, and length of hospital
stay. The unplanned reinsertion of a nasogastric tube was recorded.
The data were extracted independently by two of the authors (SJL and
PAS), checked for consistency by another author (ST), and sent to
the trialists for review.
From each study we collected data on the site of surgery, whether
an intestinal anastomosis was formed, whether the pathology was
benign or malignant, the type of feed used, and the method of
administration of the feed. The site of surgery was classified as
pancreatic, hepatobiliary, upper gastrointestinal (proximal to the
jejunum), or lower gastrointestinal (distal to the duodenum).
Outcomes potentially related to feeding included anastomotic
dehiscence, infection of any type, wound infection, pneumonia,
intra-abdominal abscess, vomiting, mortality, and length of hospital
stay. The unplanned reinsertion of a nasogastric tube was recorded.
The data were extracted independently by two of the authors (SJL and
PAS), checked for consistency by another author (ST), and sent to
the trialists for review.
Assessment of methodological quality![]() Two of us (ST and ME) independently assessed the two dimensions of
methodological quality that empirically have been shown to be
associated with biased estimates of treatment effects: adequacy of
concealment of allocation to treatment groups and double blinding. 16 17
Differences in assessment were resolved by consensus.
Two of us (ST and ME) independently assessed the two dimensions of
methodological quality that empirically have been shown to be
associated with biased estimates of treatment effects: adequacy of
concealment of allocation to treatment groups and double blinding. 16 17
Differences in assessment were resolved by consensus.
Analysis![]() We combined results from individual studies on the relative risk
scale using fixed effects meta-analysis.18 Data
on length of hospital stay were pooled with non-standardised mean differences.
We used a
We combined results from individual studies on the relative risk
scale using fixed effects meta-analysis.18 Data
on length of hospital stay were pooled with non-standardised mean differences.
We used a ![]() 2
test to test for homogeneity of relative risks. We used funnel plots
to determine the presence of publication bias and related biases and
performed a statistical test of funnel plot asymmetry.19 In
a sensitivity analysis we excluded data from patients who did not
have an intestinal anastomosis from analyses of anastomotic dehiscences.
These patients had abdominoperineal resections or stoma creations.
In a planned subgroup analysis we examined whether the risk of
anastomotic dehiscence differed according to whether the anastomosis
was proximal or distal to the site of feeding. Results are presented
as relative risks (95% confidence intervals). All analyses were
performed with Stata version 6.0 (StataCorp, College Station,
Texas).
2
test to test for homogeneity of relative risks. We used funnel plots
to determine the presence of publication bias and related biases and
performed a statistical test of funnel plot asymmetry.19 In
a sensitivity analysis we excluded data from patients who did not
have an intestinal anastomosis from analyses of anastomotic dehiscences.
These patients had abdominoperineal resections or stoma creations.
In a planned subgroup analysis we examined whether the risk of
anastomotic dehiscence differed according to whether the anastomosis
was proximal or distal to the site of feeding. Results are presented
as relative risks (95% confidence intervals). All analyses were
performed with Stata version 6.0 (StataCorp, College Station,
Texas).
|
|
Results |
Characteristics of trials, patients, and
interventions
We identified 13 randomised controlled trials, all of which
were published in English. 13 20-31 We
excluded two of these trials because no information on relevant
outcomes was given, and attempts to obtain unpublished data from the
authors were unsuccessful. 30 31
Additional unpublished data were obtained for six of the studies. 21 22 24-26 29 The
earliest study was published in 197920;
however, most were published between 1995 and 1998.
Patients had a wide variety of
gastrointestinal conditions. Table 1 gives details of the
11 trials that we included. Six studies included only lower
gastrointestinal surgery. 13 21 22 25 27 29 Six
studies did not state the underlying pathology of the study participants,
the five remaining trials included both benign and malignant
conditions. Thirty one patients from five trials underwent
abdominoperineal resections or stoma creation and had no
anastomosis. 20
22 24 25 27 In six
studies patients in the intervention group were fed directly into
the small bowel, and in five studies patients were fed orally. In
four studies feeding was started within six hours after surgery. 13 23 24 28 In the
nine remaining studies it was started within 24 hours. One
trial used a placebo feed (water).24
|
Methodological quality of trials
Reporting on concealment of allocation of treatment and blinding was
poor. In three trials allocation was concealed with sealed
envelopes, 23
24 26 and
one trial used an open table with random numbers,20 but in
the remaining studies the exact method of randomisation was unclear.
In the study by Heslin et al the outcomes were assessed by "a physician
not associated with the surgical team."26 In all
other studies outcome assessment was probably open, although this
was explicitly stated in only one report.28
Outcomes
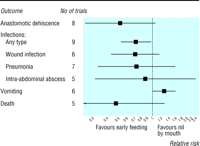
The effects of early feeding on anastomotic dehiscence, infections,
vomiting, and mortality are detailed in table 2 and summarised in the
figure. Occurrence of anastomotic dehiscence was reported in eight
of the 11 trials. Table 3 gives detailed
information on the number of events![]() for example, the risk of dehiscence ranged from 2%
(2/95) to 7% (2/30) in early feeding groups and from 1% (1/81) to
25% (4/16) in control groups. Seven trials showed that early feeding
led to a reduction in risk of anastomotic dehiscence (table 2) with a combined
relative risk of 0.53 (95% confidence interval 0.26 to
1.08, P=0.080) and no evidence of heterogeneity between studies
(
for example, the risk of dehiscence ranged from 2%
(2/95) to 7% (2/30) in early feeding groups and from 1% (1/81) to
25% (4/16) in control groups. Seven trials showed that early feeding
led to a reduction in risk of anastomotic dehiscence (table 2) with a combined
relative risk of 0.53 (95% confidence interval 0.26 to
1.08, P=0.080) and no evidence of heterogeneity between studies
(![]() 2=2.10,
P=0.96). Results were similar when 31 patients in whom no
anastomosis had been formed were excluded from the denominator of
five trials (combined relative risk 0.54, 0.26 to 1.09). 20 22 24 25 27 There
was little evidence that results differed between the two studies in
which the anastomosis was known to be proximal to the site of
feeding 26
28 and
the six trials in which it was distal (P=0.42 for interaction).
2=2.10,
P=0.96). Results were similar when 31 patients in whom no
anastomosis had been formed were excluded from the denominator of
five trials (combined relative risk 0.54, 0.26 to 1.09). 20 22 24 25 27 There
was little evidence that results differed between the two studies in
which the anastomosis was known to be proximal to the site of
feeding 26
28 and
the six trials in which it was distal (P=0.42 for interaction).
|
|
|
The risk of any type of infection was
reported in all but two trials (table 2). 21 28 Absolute
risks ranged from 3% (1/29) to 30% (29/97) in the early feeding
groups and from 5% (4/81) to 47% (14/30) in the control groups
(table 3). The
combined relative risk was 0.72 (0.54 to 0.98), indicating
a significant (P=0.036) reduction in the risk of infection, with
little evidence of heterogeneity between trials (![]() 2=10.7, P=0.22). Similar reductions were
observed for wound infection and pneumonia (figure). There was an
increase in the risk of vomiting among patients fed early
(1.27, 1.01 to 1.61, P=0.045). Absolute risks ranged
from 21% (17/80) to 50% (15/30) in the early feeding groups and from
14% (11/81) to 57% (17/30) in the control groups (table 3). When nasogastric
tubes were not placed routinely at the time of surgery the rate of
placement because of nausea and vomiting was higher in patients fed
early (1.21, 0.73 to 1.99, P=0.46).
2=10.7, P=0.22). Similar reductions were
observed for wound infection and pneumonia (figure). There was an
increase in the risk of vomiting among patients fed early
(1.27, 1.01 to 1.61, P=0.045). Absolute risks ranged
from 21% (17/80) to 50% (15/30) in the early feeding groups and from
14% (11/81) to 57% (17/30) in the control groups (table 3). When nasogastric
tubes were not placed routinely at the time of surgery the rate of
placement because of nausea and vomiting was higher in patients fed
early (1.21, 0.73 to 1.99, P=0.46).
Mortality was reported in all but two
studies, 21
28 but
deaths occurred in only five (table 2). When reported,
death occurred in hospital except for in one study in
which 30 day mortality was reported.24
Mortality ranged from none to 7% (2/30) in the early feeding groups
and 13% (4/30) in the control groups (table 3). There were four
deaths in the early feeding groups compared with 10 deaths in
control groups (relative risk 0.48, 0.18 to
1.29, P=0.15).
Length of hospital stay was reported in
all 11 studies. When combining data for meta-analysis we estimated the
mean length of stay from the median for one trial.20 We
estimated standard deviations (SD) by dividing ranges by factor
4 for two trials. 20
21 The
mean length of stay ranged from 6.2 days to 14.0 days in early feeding
groups and from 6.8 days to 19.0 days in control groups. Combined
results showed a significant reduction by 0.84 day (0.36 to
1.33 days, P=0.001), with some evidence of heterogeneity between studies
(![]() 2=16.2,
P=0.094). Results were similar when we excluded the two trials with
incomplete data. 20
21
2=16.2,
P=0.094). Results were similar when we excluded the two trials with
incomplete data. 20
21
Two major complications of feeding were
reported in patients fed via jejunostomies: one broke and migrated into the abdomen26 and
one left a prolonged fistula after it was removed.26
Funnel plots
We examined funnel plots for all nine outcomes (the seven shown in
table 2 plus length
of stay and replacement of nasogastric tubes). There was no clear
evidence of asymmetry in any of these plots (P>0.10 by regression
test19),
except for mortality (P=0.068).
|
|
Discussion |
This meta-analysis yielded three principal findings. Firstly,
there does not seem to be a clear advantage in keeping patients nil
by mouth after elective gastrointestinal resection. Secondly, in
these patients early feeding may be beneficial. Thirdly, we believe
these results indicate the necessity for an adequately powered
clinical trial to assess early enteral feeding in patients undergoing
elective gastrointestinal surgery.
Complications after operation
Anastomotic dehiscence is a major complication of gastrointestinal
surgery with considerable morbidity and mortality.32 The
combined estimate failed to reach conventional levels of significance but
indicates that early feeding may reduce the risk of dehiscence. Reporting
on factors that could have modified the effect of early feeding,32-34 such
as the experience of the surgeon, whether the resections were from
the large or small bowel, the length of the operation time,
postoperative pain control, the use of antibiotics, and the success
of the operation, was incomplete. Furthermore, the definition of
dehiscence varied between trials. However, the estimated effect in
eight out of nine studies that reported anastomotic dehiscence
indicated benefit and was similar among patients fed proximally or
distally to their anastomosis.
A significant relative reduction in the
risk of infection of any type was observed for patients receiving early enteral
nutrition, with the greatest reduction seen in the frequency of
wound infections. In most of the trials assessed infections were not
clearly defined. In absolute terms results were heterogeneous, with
the number of patients who would need to be treated to prevent one
infection of any type ranging from three24 to 58.26
Length of hospital stay
The length of hospital stay after surgery was reduced in eight of
the eleven studies. Overall the reduction corresponds to about one
day, which is economically important. Reduction in complication
rates may explain this observation, as might a faster return of
gastrointestinal function. Early postoperative feeding after
non-gastrointestinal surgery has also been shown to reduce length of
stay in hospital. 35
36
Although not significant, the direction of effect suggested a
reduced risk of postoperative death among patients who received
early enteral feeding in all five studies in which mortality
occurred. There were insufficient data to comment on the causes of
death. Reductions in mortality tended to be larger in smaller
studies, which may be due to chance, publication bias, or lower
methodological quality of smaller studies.19 Mortality
was one out of nine outcomes examined, and no evidence of small
study bias was evident for the other outcomes. The association found
for mortality was probably a chance finding.
Statistical quality
The 11 randomised trials identified were clinically heterogeneous
and most of them were small and of doubtful methodological quality.
Combination trials that differ in terms of underlying condition,
operation, and intervention may be inappropriate. However, we were
interested in the pragmatic comparison of early versus deferred
feeding strategies after gastrointestinal surgery and not in
differences between feed types or specific routes of feeding. It is
noteworthy that the effect of early nutrition seemed to be homogeneous
across a set of trials that were clearly heterogeneous in clinical
terms. Our ability to detect heterogeneity between trials, however,
was limited by the small number of trials and by the often
inadequate reporting. For example, definition of the patients'
pre-existing nutritional state and severity of underlying disease
was generally poor. The method of randomisation and blinding of
outcome assessment was also not described in sufficient detail, which
means that uncertainty regarding the methodological quality of
trials remains. In particular, the identical or closely similar number
of patients in comparison groups in these trials must be of concern.37 This
could occur only if blocked randomisation with a small block size
had been used. Blocked or stratified randomisation, however, was
mentioned in only two trials. 24 28
Conclusion
There is little evidence from these trials that keeping patients nil
by mouth is beneficial after elective gastrointestinal resection.
Although the data are clearly insufficient to conclude that early
feeding is of proved benefit, we believe that there is a good case
for an adequately powered clinical trial to assess early enteral
feeding in such patients. With anastomotic dehiscence as the primary
end point, such a trial would need to enrol about 1000 patients
in each arm and would therefore involve several centres.
|
|
Acknowledgments |
We thank the authors who provided
additional data: R Beier-Holgersen, B Stewart, S Wexner, J E Fischer, M F
Brennan, and H Ortiz. We also thank the following manufacturers of
enteral feeds for assisting in the collection of data: Nutricia,
Fresenius Kabi, Novartis Nutrition UK, Ross Products, Clintec
Nutrition, Nestlé (Clinical Nutrition), SHS International, Mead
Johnson Nutrition.
Contributors: The data were extracted independently by SJL and PAS
and checked for consistency by ST. ST and ME assessed the two dimensions of
methodological quality. SJL wrote to all the trialists and feed companies
requesting additional data and coordinated the writing of the paper. ME advised
on the literature search and performed quality assessments and statistical
analyses. All the authors contributed to the writing of the final draft of the
manuscript. SJL is guarantor for the paper.
|
|
Footnotes |
Funding: None.
Competing interests: None declared.
|
|
References |
|
1. |
Catchpole BN. Smooth muscle and the
surgeon. Aust N Z J Surg 1989;
59: 199-208 |
|
2. |
Moss G. Maintenance of gastrointestinal
function after bowel surgery and immediate enteral full nutrition. II.
Clinical experience, with objective demonstration of intestinal absorption
and motility. J Parenter Enteral Nutr
1981; 5: 215-220 |
|
3. |
McCarter MD, Gomez ME, Daly JM. Early
postoperative enteral feeding following major upper gastrointestinal surgery.
J Gastrointest Surg 1996; 1:
278-285 |
|
4. |
McWhirter JP, Pennington CR. Incidence
and recognition of malnutrition in hospital. BMJ
1994; 308: 945-948 |
|
5. |
Hill GL, Pickford I, Young GA, Schorah
CJ, Blackett RL, Burkinshaw L, et al. Malnutrition in surgical patients: an
unrecognised problem. Lancet
1977; i: 689-692 |
|
6. |
Lennard-Jones JE. A positive approach to nutrition as a treatment.
London: King's Fund Centre, 1992. |
|
7. |
The Veterans Affairs Total Parenteral
Nutrition Cooperative Study Group. Perioperative total parenteral nutrition
in surgical patients. N Engl J Med
1991; 325: 525-522 |
|
8. |
Uden P, Blomquist P, Jiborn H,
Zederfeldt B. Impact of long-term relative bowel rest on conditions for
colonic surgery. Am J Surg
1988; 156: 381-385 |
|
9. |
Irvin TT, Hunt TK. Effect of
malnutrition on colonic healing. Ann Surg
1974; 180: 765-772 |
|
10. |
Ward MW, Danzi M, Lewin MR, Rennie MJ,
Clark CG. The effects of subclinical malnutrition and refeeding on the
healing of experimental colonic anastomoses. Br
J Surg 1982; 69: 308-310 |
|
11. |
Goodlad RA, Al-Mukhtar MY, Ghatei MA,
Bloom SR, Wright NA. Cell proliferation, plasma enteroglucagon and plasma
gastrin levels in starved and refed rats. Virchows
Arch B Cell Pathol Incl Mol Pathol 1983; 43: 55-62 |
|
12. |
Moss G, Greenstein A, Levy S, Bierenbaum
A. Maintenance of GI function after bowel surgery and immediate enteral full
nutrition. I. Doubling of canine colorectal anastomotic bursting
pressure and intestinal wound mature collagen content. Clinical experience,
with objective demonstration of intestinal absorption and motility. J Parenter Enteral Nutr 1980; 4: 535-538 |
|
13. |
Schroeder D, Gillanders L, Mahr K, Hill
GL. Effects of immediate postoperative enteral nutrition on body composition,
muscle function, and wound healing. J
Parenter Enteral Nutr 1991; 15: 376-383 |
|
14. |
Moore FA, Moore EE, Jones TN, McCroskey
BL, Peterson VM. TEN versus TPN following major abdominal trauma |
|
15. |
Windsor AC, Kanwar S, Li AG, Barnes E,
Guthrie JA, Spark JI, et al. Compared with parenteral nutrition, enteral
feeding attenuates the acute phase response and improves disease severity in
acute pancreatitis. Gut 1998;
42: 431-435 |
|
16. |
Schulz KF, Chalmers I, Hayes RJ, Altman
DG. Empirical evidence of bias. Dimensions of methodological quality
associated with estimates of treatment effects in controlled trials. JAMA 1995; 273: 408-412 |
|
17. |
Moher D, Pham B, Jones A, Cook DJ, Jadad
AR, Moher M, et al. Does quality of reports of randomised trials affect
estimates of intervention efficacy reported in meta-analyses? Lancet 1998; 352: 609-613 |
|
18. |
Deeks JJ, Altman DG, Bradburn MJ.
Statistical methods for examining heterogeneity and combining results from
several studies in meta-analysis. In: Egger M, Smith DG, Altman DG, eds. Systematic reviews in health care: meta-analysis in
context. London: BMJ Publishing, 2001:285-312. |
|
19. |
Egger M, Davey Smith G, Schneider M,
Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997; 315: 629-634 |
|
20. |
Sagar S, Harland P, Shields R. Early
postoperative feeding with elemental diet. BMJ
1979; i: 293-295 |
|
21. |
Binderow SR, Cohen SM, Wexner SD,
Nogueras JJ. Must early postoperative oral intake be limited to laparoscopy? Dis Colon Rectum 1994; 37: 584-589 |
|
22. |
Reissman P, Teoh TA, Cohen SM, Weiss EG,
Nogueras JJ, Wexner SD. Is early oral feeding safe after elective colorectal
surgery? A prospective randomised trial. Ann
Surg 1995; 222: 73-77 |
|
23. |
Carr CS, Ling KD, Boulos P, Singer M.
Randomised trial of safety and efficacy of immediate postoperative enteral
feeding in patients undergoing gastrointestinal resection. BMJ 1996; 312: 869-871 |
|
24. |
Beier-Holgersen R, Boesby S. Influence
of postoperative enteral nutrition on postsurgical infections. Gut 1996; 39: 833-835 |
|
25. |
Ortiz H, Armendariz P, Yarnoz C. Is
early postoperative feeding feasible in elective colon and rectal surgery? Int J Colorectal Dis 1996; 11: 119-121 |
|
26. |
Heslin MJ, Latkany L, Leung D, Brooks
AD, Hochwald SN, Pisters PW, et al. A prospective, randomized trial of early
enteral feeding after resection of upper gastrointestinal malignancy. Ann Surg 1997; 226: 567-680 |
|
27. |
Hartsell PA, Frazee RC, Harrison JB,
Smith RW. Early postoperative feeding after elective colorectal surgery. Arch Surg 1997; 132: 518-521 |
|
28. |
Watters JM, Kirkpatrick SM, Norris SB,
Shamji FM, Wells GA. Immediate postoperative enteral feeding results in
impaired respiratory mechanics and decreased mobility. Ann Surg 1997; 226: 369-380 |
|
29. |
Stewart BT, Woods RJ, Collopy BT, Fink
RJ, Mackay JR, Keck JO. Early feeding after elective open colorectal
resections: a prospective randomized trial. Aust
N Z J Surg 1998; 68: 125-128 |
|
30. |
Ryan Jr JA, Page CP, Babcock L. Early
postoperative jejunal feeding of elemental diet in gastrointestinal surgery. Am Surg 1981; 47: 393-403 |
|
31. |
Hoover Jr HC, Ryan JA, Anderson EJ,
Fischer JE. Nutritional benefits of immediate postoperative jejunal feeding
of an elemental diet. Am J Surg
1980; 139: 153-159 |
|
32. |
Wheeler JM, Gilbert JM. Controlled intraoperative
water testing of left-sided colorectal anastomoses: are ileostomies
avoidable. Ann R Coll Surg Engl
1999; 81: 105-108 |
|
33. |
Irvin TT, Goligher JC. Aetiology of
disruption of intestinal anastomoses. Br J
Surg 1973; 60: 461-464 |
|
34. |
Thornton FJ, Barbul A. Healing in the
gastrointestinal tract. Surg Clin North Am
1997; 77: 549-573 |
|
35. |
Delmi M, Rapin CH, Bengoa JM, Delmas PD,
Vasey H, Bonjour JP. Dietary supplementation in elderly patients with
fractured neck of the femur. Lancet
1990; 335: 1013-1016 |
|
36. |
Schilder JM, Hurteau JA, Look KY, Moore
DH, Raff G, Stehman FB, et al. A prospective controlled trial of early
postoperative oral intake following major abdominal gynecologic surgery. Gynecol Oncol 1997; 67: 235-240 |
|
37. |
Schulz KF, Chalmers I, Grimes DA, Altman
DG. Assessing the quality of randomization from reports of controlled trials
published in obstetrics and gynecology journals. JAMA 1994; 272: 125-128 |
(Accepted 30 May 2001)
Edward E.
Rylander, M.D.
Diplomat American
Board of Family Practice.
Diplomat American
Board of Palliative Medicine.