Hypercoagulability Syndromes
![]() Robert H. Thomas, MD
Robert H. Thomas, MD
Hypercoagulability can be defined as the
tendency to have thrombosis as a result of certain inherited and/or acquired
molecular defects. Clinical manifestations of hypercoagulability can be
devastating and even lethal. In the past 20 years, the origin of most of these
diverse hypercoagulability syndromes has been elucidated. Currently,
hypercoagulability disorders can be correctly diagnosed in approximately 80% to
90% of patients. Defining the cause of hypercoagulability may determine the type
and duration of treatment for the associated thrombosis. The discovery of an
occult carcinoma allows for the possibility of early and possibly curative
treatment. Finding a genetic defect in coagulation allows for testing of
asymptomatic family members as well. The purpose of this review is to provide
internists with a logical approach to the identification and treatment of
hypercoagulability syndromes.
Arch Intern Med.
2001;161:2433-2439
![]()
Most commonly, thrombosis is the result of more
than one "hit." For example, patients with the factor V Leiden defect
may be asymptomatic until they start taking oral contraceptives. Patients with
antithrombin deficiency may go on without incident until they undergo a hernia
repair. Also, multiple genetic defects predispose one to thrombosis much more
than does a single defect.1, 2 Some defects are known
to be more powerful predictors than others. Therefore, hypercoagulability is
not a uniform disease process but rather a host of predisposing conditions that
may or may not be expressed as thrombosis, depending on environmental insults
and the strength and number of predisposing factors.
INDIVIDUAL SYNDROMES

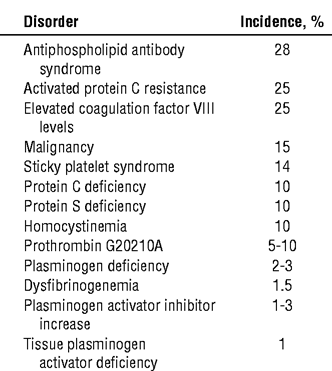
The following is a list of the disorders that
cause hypercoagulability and their approximate incidences. Since these were
derived from different studies, percentages cannot be exact. Also, incidences
may vary, depending on the ethnic backgrounds of persons in a particular
geographic area.
|
|
Antiphospholipid
Antibody Syndrome
The antiphospholipid antibody syndrome is probably the most common of the
hypercoagulable disorders. It is caused by a heterogeneous family of
immunoglobulins that bind to plasma proteins that have an affinity for
phospholipid surfaces. These antigens include B2 glycoprotein I,
prothrombin, high- and low-molecular-weight kininogens, annexin V, activated
protein C, and activated protein S. It is usually acquired and can be divided
into the lupus anticoagulant syndrome and the anticardiolipin antibody
syndrome. Both of these syndromes may be associated with other disorders, such
as collagen vascular diseases or infections, but are more often primary.
Antiphospholipid antibody syndrome can also be associated with use of the
following medications: phenytoin, quinidine, hydralazine, procainamide
hydrochloride, phenothiazines, interferon, cocaine, quinine, and the
combination product of pyrimethamine and sulfadoxine. Usually, a patient will
have one syndrome or the other but not both. Multiple mechanisms as to the
reason for hypercoagulability have been postulated, but the exact cause is
unknown at this time. The risk of thrombosis is 5.5% per year for symptomatic
patients.3
The lupus anticoagulant is directed against
phospholipids, which then causes an in vivo prolongation in the prothrombin
time (PT), partial thromboplastin time (PTT), or the Russell viper venom time.
These values do not correct with normal plasma. However, the addition of
phospholipids will correct the abnormality. Despite the prolonged coagulation
times, thrombosis is the predominant feature of this syndrome. The PT and PTT
are not sensitive enough to be used as a screening tool for the lupus
anticoagulant. Instead, the Russell viper venom time must be used. Venous
thrombosis is much more common than arterial thrombosis in these patients.4
The anticardiolipin antibody syndrome is 5 times
more common than the lupus anticoagulant syndrome.5 Antibodies can be
detected by enzyme-linked immunosorbent assay. Both IgG and IgM are associated
with thrombosis.4, 5 A total of 1% to 7% of
asymptomatic individuals have low titers of these antibodies.6 Even asymptomatic
persons have a 1% risk per year of thrombosis. This increases to 6% in those
with high titers.7 This contrasts with a
0.1% risk per year in the general population. Venous and arterial thrombi are
equally common.4 Other manifestations
include valvular abnormalities, livedo reticularis, superficial
thrombophlebitis, ulcers, adrenal hemorrhage, fetal wastage, chorea, transverse
myelopathy, and thrombocytopenia.
The treatment of patients with antiphospholipid
antibody syndrome who have had thrombosis is long-term anticoagulation until
the antibody has been absent for at least 6 months.8 The drug of choice is
low-molecular-weight heparin sodium since in 65% of patients warfarin sodium
therapy fails8 and the international
normalized ratio is unreliable in monitoring the intensity of therapy. If
warfarin must be used, an international normalized ratio target range of 3 to 4
should be sought.9 The treatment of fetal
wastage syndrome is beyond the scope of this review. There is no clear
indication for therapy in asymptomatic persons; however, aspirin therapy would
be reasonable in this population because the risk of thrombosis is higher than
normal. Other treatments, such as corticosteroids, cyclophosphamide, and plasma
exchange, have been used for severely symptomatic disease, but their roles in
routine management are not well established.
Activated Protein C Resistance
Activated protein C resistance (eg, factor V Leiden) is the most common
inherited disorder that causes hypercoagulability. Factor V Leiden is present
in 5% of whites but virtually absent in Africans and Asians. However, 1% of
African Americans have the mutation, reflecting racial mixing.10 It results from a
point mutation in the factor V gene, which causes the substitution of glutamine
for arginine at position 506.11 (Several other rare
factor V gene mutations that can lead to activated protein C resistance have
also been described.12-14) Consequently, 1
of 3 activated protein C cleavage sites is lost. The result is an impaired
inactivation of factor V by activated protein C. Venous thromboses and fetal
wastage may occur. It is not an important risk factor for arterial disease
except in the presence of smoking or other known risk factors.15, 16 Those with factor V
Leiden have a 2- to 3-fold risk for venous thrombosis compared with healthy
subjects. The risk in homozygotes is 80-fold.17 Heterozygous factor V
Leiden is, therefore, a relatively mild risk factor for thrombosis.15, 18 The annual rate of
thrombosis is 0.28%.19 Six percent of
patients will have a thrombosis by the age of 65 years.20 Sixty percent of
patients who experience thrombosis have a predisposing event, such as oral
contraceptive use or pregnancy.21 The presence of this
mutation does not appear to affect life expectancy, and many patients will
remain asymptomatic. Therefore, patients with no history of thrombosis should
not be treated prophylactically with long-term anticoagulation.22 Functional tests for
activated protein C resistance should be used to screen for the disorder, and
positive results should be confirmed with polymerase chain reaction for the
genetic mutation. However, patients with phenotypic resistance to activated
protein C have an increased risk of thrombosis even if it is not due to factor
V Leiden.20 Functional
tests may still be performed while patients undergo anticoagulation.
Elevated Coagulation Factor
VIII Levels
Elevated coagulation factor VIII levels appear to be nearly as common a risk
factor for thrombosis as factor V Leiden. The Leiden Thrombophilia Study found
an 11% incidence in healthy controls and a 25% incidence in patients with
venous thrombosis. The odds ratio for thrombosis was 4.8 for subjects with levels
greater than 150 IU/dL vs those with levels less than 100 IU/dL.23, 24 For every 10-IU/dL
rise in levels, the risk for a single episode of deep venous thrombosis (DVT)
increases 10% and the risk for recurrent DVT increases 24%.25 Levels of coagulation
factor VIII are not elevated because of the acute-phase reaction but appear to
be constitutively increased in most patients with thrombosis, since coagulation
factor VIII levels are elevated independently of C-reactive protein and
fibrinogen, and 94% of patients continue to have high levels throughout
long-term follow-up.26, 27 Pregnancy and oral
contraceptive use may also raise levels. The use of oral contraceptives in
patients with increased coagulation factor VIII levels raises the risk of
thrombosis 10-fold over patients with neither risk factor.28 The genetic basis for
increased coagulation factor VIII levels is not well understood at this time;
however, one small study25 showed high
concordance rates for first-degree adult family members.
Malignancy
Cancer is the second most common acquired cause of hypercoagulability,
accounting for 10% to 20% of spontaneous DVTs. Indeed, 15% of patients with
cancer have clinical thromboses and about 50% have thromboses on autopsy.29 Cancer not only
causes hypercoagulability but may also produce endothelial injury and venous
stasis. Hypercoagulability is especially frequent in mucin-secreting
adenocarcinomas, brain tumors, acute promyelocytic leukemia, and
myeloproliferative disorders.
Arterial thrombosis is much less common than
venous thrombosis and is most often the result of nonbacterial thrombotic
endocarditis or disseminated intravascular coagulation. Ninety percent of
patients with cancer have clotting abnormalities, such as increased fibrinogen,
clotting factors, fibrin degradation products, and platelets.30, 31 Overt disseminated
intravascular coagulation is rare. There is no consensus as to the value of
measuring coagulation markers in predicting thrombosis in individual patients
with cancer.
Some cancers underlying spontaneous DVT are
occult, early stage, and curable. However, there is no proof that aggressive
diagnostic testing leads to improvement in survival. Most experts recommend a
thorough history and physical examination, routine blood tests, chest x-ray
examination, urinalysis, and age- and sex-specific screening, such as
prostate-specific antigen, Papanicolaou smear, lower endoscopy, mammography,
and fecal occult blood testing. Suspicious findings should be aggressively
evaluated. In addition, patients without evidence of cancer should be followed
up closely for the ensuing 2 years, during which time virtually all occult
cancers will become clinically apparent.
The initial treatment of thromboses is the same
as in patients without cancer. However, treatment should be continued
indefinitely until the patient is cured of the malignancy and is no longer
receiving chemotherapy. If anticoagulation is contraindicated as with cerebral
or pericardial metastases, primary brain tumors, or severe thrombocytopenia, an
inferior vena cava filter may be placed. Long-term treatment may be with
low-molecular-weight heparin or warfarin, although anecdotal evidence suggests
that heparin may lead to fewer thrombotic recurrences than warfarin.32 Certainly, warfarin
failure should lead to a switch to heparin. If heparin fails, an inferior vena
cava filter should then be placed. Thrombolytic agents should only be used in
patients with cancer who have a good prognosis and either pulmonary embolism
with hemodynamic compromise or severe iliofemoral thrombosis of less than 4
days' duration. Of course, aggressive DVT prophylaxis with low-dose
subcutaneous heparin or low-molecular-weight heparin (depending on severity and
number of risk factors) should be carried out in patients with cancer who are
hospitalized, immobilized, or undergoing surgery.
Sticky Platelet Syndrome
The sticky platelet syndrome is an autosomal dominant disorder that results in
platelets that are hyperaggregable to epinephrine and/or adenosine diphosphate.
Venous or arterial thrombosis may occur.33 Episodes are more
common during emotional stress. Retinal vascular thrombosis appears to be
associated with this entity. Fetal wastage may also occur. It is diagnosed with
platelet aggregation studies. Treatment is with low-dose aspirin (81 mg). If
platelet aggregability does not normalize, aspirin, 325 mg, may be tried.34 If there is still no
response, then clopidogrel (an adenosine diphosphate receptor antagonist
similar to but better tolerated than ticlopidine hydrochloride) may be used.
Protein C Deficiency
Protein C deficiency is an autosomal dominant trait that may be caused by a
decrease in absolute levels of protein C or a decrease in its function.
Deficiency of protein C occurs in 1 of 250 controls.35 Protein C is made in
the liver and is vitamin K dependent. It acts to inactivate factor V and factor
VIII:C. It requires factor S as a cofactor and is activated by thrombin, when
thrombin is bound to thrombomodulin.
In families with thromboses and protein C
deficiency, thromboses begin in the late teens.36 Seventy-five percent
of affected individuals will have 1 or more events.38 The relative risk is
7.3.18 The annual
incidence is 1%.19 Seventy percent of
episodes are spontaneous.37 Both DVT and
pulmonary embolism are the most common manifestations. Superficial
thrombophlebitis is also common.38 Arterial events are
rare.
The optimal time to investigate is at least 10
days after warfarin therapy is stopped, since both warfarin and acute
thrombosis decrease protein C levels. Levels below 55% of normal are likely to
be genetically deficient; 55% to 65% is borderline. Abnormal results should
always be repeated for confirmation and family studies performed.39
The short-term management of thrombosis is with
heparin or low-molecular-weight heparin. Warfarin may be used for long-term
treatment; however, doses should be started low and titrated upward slowly only
after heparin is therapeutic because of the risk of warfarin necrosis.40 In fact, one third of
patients with warfarin necrosis have an underlying protein C deficiency.
Protein S Deficiency
Protein S is vitamin K dependent and is synthesized by hepatocytes and
megakaryocytes. It acts as a cofactor for protein C. Fifty percent circulates
free and 50% circulates bound to C4b binding protein. Deficiency is transmitted
autosomally dominant and can be quantitative or qualitative.
Seventy-four percent of patients develop DVT;
72% develop superficial thrombophlebitis.41 The relative risk of
thrombosis is 8.5.18 The annual incidence
is 1%19; 56% of
episodes are spontaneous. Arterial events are uncommon. One half of patients
who develop thromboses do so by the age of 25 years.41
Short-term therapy is standard. Long-term
therapy is with warfarin or low-molecular-weight heparin. Since warfarin
necrosis may occur, therapy should be started with warfarin at low doses and
increased slowly after heparin has been administered. While the patient is
undergoing warfarin therapy, protein C and S levels decrease by 50% within 48
hours and then increase to 70% of usual levels after 2 weeks. Therefore, levels
below 60% of normal while taking warfarin in the long term are suspicious for
deficiency.8
Homocystinemia
Elevated levels of homocysteine are known to be a risk factor for arterial and
venous thrombosis and fetal wastage. Homocysteine is an intermediate of
methionine metabolism and, therefore, elevated levels may result from
cystathionine ![]() -synthase
deficiency, homozygous expression of the thermolabile form of
methylenetetrahydrofolate reductase, or from B12 or folic acid
deficiency. Mild-to-moderate increases in homocysteine occur in 5% to 10% of
the population.42 The relative risk of
thrombosis is 2.6.43
-synthase
deficiency, homozygous expression of the thermolabile form of
methylenetetrahydrofolate reductase, or from B12 or folic acid
deficiency. Mild-to-moderate increases in homocysteine occur in 5% to 10% of
the population.42 The relative risk of
thrombosis is 2.6.43
Elevated homocysteine levels are thought to
cause thromboses via several mechanisms, including (1) decreased protein C
activation, (2) increased factor V activity, (3) induction of endothelial cell
tissue factor activity, (4) inhibition of thrombomodulin expression and
activation, (5) decreased antithrombin activity, and (6) enhanced affinity of
lipoprotein(a) and fibrin.44-46
Measurement of homocysteine levels is not well
standardized, and acute thrombosis may raise homocysteine levels. Dietary
supplementation with vitamin B6, B12, and folic acid can
lower homocysteine levels.47 However, reduction of
homocysteine levels has not been shown to reduce thrombotic complications.
Folate supplementation (400 µg/d) may decrease levels by 30% to 42%. B12
supplementation (100 µg/d) may decrease levels by 15%. B6
supplementation (3 µg/d) only reduces levels if there is a preexisting
deficiency. Thrombosis is treated in standard fashion in addition to vitamin
supplementation.
Antithrombin Deficiency
Antithrombin is made in the liver and endothelial cells. It inactivates
thrombin and other serine proteases. Deficiency is an autosomal dominant
disorder and occurs in 1 of 5000 healthy blood donors.48 The protein may be
absent or dysfunctional. The normal concentration is 150 µg/mL. Thrombosis may
occur at less than 75% of this amount. Patients may present with DVT or
pulmonary embolism. Mesenteric vessels appear to be particularly susceptible.
Arterial events are rare. Fifty percent of patients are asymptomatic.
Thromboses occur early in life, with two thirds of patients presenting by the
age of 35 years. Forty percent of thromboses are spontaneous.49 The relative risk of
thrombosis is 8.1,18 and the annual
incidence of thrombosis is 1%.19
Acute thrombosis, heparin, and other systemic
diseases may decrease antithrombin levels.8 Warfarin may raise
deficient levels into the normal range.50 Therefore, low levels
in a patient during acute thrombosis or while taking heparin should be
confirmed when the patient is not undergoing therapy. Likewise, normal levels
while the patient is taking warfarin should be confirmed when the patient is
not undergoing therapy.
Treatment of acute thrombosis is with
low-molecular-weight heparin because deficiency may cause resistance to
unfractionated heparin.51 In fact, heparin
resistance may be a clue to the presence of this deficiency. Lifelong therapy
should be considered for spontaneous or recurrent thromboses. Prophylactic
treatment of asymptomatic individuals is controversial but usually is limited
to high-risk situations, such as pregnancy or surgery. Antithrombin concentrate
may be considered for situations in which both thrombosis and bleeding may
occur, such as labor and delivery, where anticoagulation might be
contraindicated.52
Dysfibrinolysis
There are 5 major forms of dysfibrinolysis: (1) congenital plasminogen
deficiency, (2) tissue plasminogen activator deficiency, (3) increased
plasminogen activator inhibitor, (4) congenital dysfibrinogenemia, and (5)
factor XII deficiency. Long-term treatment may be with warfarin or
low-molecular-weight heparin for all patients.
Congenital plasminogen deficiency is a rare
autosomal dominant disorder caused by either absent or dysfunctional
plasminogen. Clinically, it mimics protein C and S deficiencies. Symptoms
usually begin in the late teens. Most commonly, it presents with DVT or
pulmonary embolism. Arterial events are uncommon. Events usually occur when
plasminogen levels are less than 40% of the normal values. The results of
routine coagulation studies are normal.53 Treatment is
standard.
Congenital deficiency of tissue plasminogen
activator and congenital increases of plasminogen activator inhibitor are
exceedingly rare. Acquired abnormalities are more common. They may occur with
diabetes mellitus, inflammatory bowel disease, and coronary atherosclerosis.54-56
Most congenital dysfibrinogenemias occur in
asymptomatic individuals (55% of patients) or cause mild hemorrhagic disorders
(20%). Only 20% are associated with thrombosis.57 Venous thrombosis is
most common but arterial events may occur. They are usually autosomal dominant.
They may be detected with abnormal thrombin times or reptilase clotting times.
Treatment of thrombosis consists of heparin or low-molecular-weight heparin
followed by warfarin.
Factor XII deficiency is inherited in autosomal
dominant fashion. It is involved in plasmin generation. Thus, patients will
have a prolonged PTT, yet have a thrombotic diathesis. Arterial and venous
thromboses and fetal wastage are common. Approximately 8% of deficient subjects
develop thromboses.58 Factor XII deficiency
should be suspected when a patient with thrombosis has a prolonged PTT that
corrects with the addition of normal plasma. A factor XII assay should then be
performed. Treatment is with low-molecular-weight heparin followed by warfarin
or continuation of low-molecular-weight heparin. Standard unfractionated
heparin should not be used because of difficulties in monitoring the PTT.
Prothrombin G20210A
Prothrombin G20210A mutation is a relatively recently discovered defect in
which there is a G to A transition at nucleotide position 20210. This mutation
increases prothrombin activity and levels.59 It is found in 2.3%
of healthy controls. The incidence is twice as high in people from southern
Europe than from northern Europe, and it is rare in Africans and Asians.60 It may be detected
through DNA analysis. At this time, it must be considered a very mild risk
factor for venous and arterial thrombosis. The relative risk is approximately 2
to 3 times that of individuals without the mutation.61-63
Other Hypercoagulable Syndromes
Heparin cofactor II inhibits thrombin by mimicking the cleavage sites of
thrombin and forming a stable complex with it, thus acting as a
"suicide" substrate. Deficiency is rare and could theoretically cause
thrombotic potential, but its exact role is controversial. Heparin is effective
in the presence of heparin cofactor II deficiency.
Tissue factor pathway inhibitor is a plasma
component that binds and inhibits factor Xa directly. This complex then binds
to the tissue factor–factor VIIa complex, blocking its activity as well.
Unstimulated plasma levels do not appear to be related to thrombosis. However,
plasma levels measured 10 minutes after intravenous heparin, 7500 U, is
administered correlate with venous thrombosis.64 The role of tissue
factor pathway inhibitor and its incidence in thrombophilia are currently
unknown.
Thrombomodulin mutations have also been
implicated in thrombophilia but prevalence and degree of risk are unknown.65
The Wein-Penzing defect is an extremely rare
deficiency of the lipoxygenase metabolic pathway that results in the
compensatory increase of the cyclooxygenase pathway and, therefore, elevated
thromboxane levels. Thus, platelets are in a state of increased activation.
INVESTIGATION OF HYPERCOAGULABILITY

Various clinical features should suggest
hypercoagulability, including thrombosis at an early age (<50 years), family
history of thrombosis, recurrent idiopathic thrombosis, thrombosis at an
unusual site (except for effort-related upper extremity DVT66), spontaneous
thrombosis or only mild provocation, unexplained spontaneous abortions, massive
thrombosis, and warfarin-induced skin necrosis. Information from the history
and physical examination determines the likelihood of the underlying disorder.
For example, a young patient who presents with a strong family history of
thrombosis suggests a genetic disorder. A patient with systemic lupus
erythematosus is likely to have the antiphospholipid antibody syndrome. An
older patient with weight loss, early satiety, and epigastric pain is likely to
have a gastric carcinoma.
All patients should have a complete blood cell
count performed, including platelets, to exclude myeloproliferative disorders.
Abnormalities in PT and PTT suggest either the lupus anticoagulant or factor
XII deficiency. Antiphospholipid antibodies should be obtained. Screening for
cancer as outlined earlier should be performed. Patients in whom this
evaluation is negative and all patients with a positive family history of
thrombosis should undergo testing for the common genetic disorders (Table 1).
Table 2
provides a list of the approximate costs of the various tests for
hypercoagulability. Laboratory investigation for these disorders is generally
unreliable during acute thrombosis and while undergoing anticoagulant therapy.
Thus, studies are optimally performed while the patient is not taking
anticoagulants and is in the asymptomatic state. If tests are performed while
the patient is taking anticoagulants, knowledge of the alteration of the
individual factors by the specific anticoagulant is essential. Depending on the
level of suspicion for a genetic defect, referral to a hematologist for testing
of the rarer defects may be indicated if the prior workup is unrewarding. A
useful mnemonic for the common causes of hypercoagulability is CALMSHAPES:
protein C deficiency, Antiphospholipid antibody syndrome, factor
V Leiden; Malignancy, protein S deficiency, Homocystinemia, Antithrombin
deficiency, Prothrombin G20210A,
increased factor VIII (Eight), Sticky platelet syndrome.
TREATMENT

When considering a patient for indefinite
therapy, many factors must be considered: (1) the number, site, and severity of
thromboses; (2) spontaneous vs provoked thrombus; (3) the sex and lifestyle of
the patient; (4) the strength of the predictive value for thrombosis of the
particular hypercoagulable disorder; (5) the compliance of the patient; and (6)
the patient's personal value construct.
Few guidelines exist for indefinite therapy in
hypercoagulable patients. A recent study67 that favored
indefinite therapy in anyone with an unprovoked thrombosis was terminated
prematurely. Bauer68 divides patients with
hereditary defects into 2 groups: high risk (![]() 2
spontaneous episodes, 1 spontaneous life-threatening thrombosis, 1 thrombosis
at an unusual site, or 1 thrombosis in the presence of >1 defect) and
moderate risk (asymptomatic individuals or 1 thrombosis in response to a
prothrombotic stimulus). In the high-risk group, he recommends indefinite
anticoagulation. In the moderate-risk group, he recommends vigorous prophylaxis
only for high-risk situations. Since no long-term studies have been performed
comparing lifetime anticoagulation treatment with short-term anticoagulation
therapy, definitive recommendations cannot be made at this time. Until these
studies are performed, Figure 1
may be used as a guide to the evaluation and management of hypercoagulable
disorders.
2
spontaneous episodes, 1 spontaneous life-threatening thrombosis, 1 thrombosis
at an unusual site, or 1 thrombosis in the presence of >1 defect) and
moderate risk (asymptomatic individuals or 1 thrombosis in response to a
prothrombotic stimulus). In the high-risk group, he recommends indefinite
anticoagulation. In the moderate-risk group, he recommends vigorous prophylaxis
only for high-risk situations. Since no long-term studies have been performed
comparing lifetime anticoagulation treatment with short-term anticoagulation
therapy, definitive recommendations cannot be made at this time. Until these
studies are performed, Figure 1
may be used as a guide to the evaluation and management of hypercoagulable
disorders.
CONCLUSIONS

The evaluation and treatment of a patient
suspected of having hypercoagulability cannot be generalized at this time. The
clinician must consider many patient factors with statistical probabilities to
determine what conditions should be investigated. When a hypercoagulable
syndrome is diagnosed, further judgment must be exercised to then decide the
best course of treatment. Oversimplifications on the evaluation and treatment
of hypercoagulable syndromes are not helpful and may result in harm to
individual patients.
Author/Article Information
![]()
From the Department of General Medicine, University of Miami School of
Medicine, Miami, Fla.
Corresponding author and reprints: Robert H. Thomas, MD, Department of General
Medicine, University of Miami School of Medicine, 1475 NW 12th Ave, Third
Floor, Miami, FL 33136 (e-mail: [log in to unmask]).
Accepted for publication April 9, 2001.
I would like to thank Barry Materson, MD, for
his excellent advice and assistance in editing the manuscript.
REFERENCES

1.
Salomon O, Steinberg DM, Zivelin A, et al.
Single and combined prothrombotic factors in patients with idiopathic venous
thromboembolism: prevalence and risk assessment.
Arterioscler Thromb Vasc Biol.
1999;19:511-518.
MEDLINE
2.
DeStefano U, Martinelli I, Mannucci P, et al.
The risk of recurrent deep venous thrombosis among heterozygous carriers of
both factor V Leiden and the G20210A prothrombin mutation.
N Engl J Med.
1999;341:801-806.
MEDLINE
3.
Finazzi G, Brancaccio U, Moia M, et al.
Natural history and risk factors for thrombosis in 360 patients with
antiphospholipid antibodies: a four-year prospective study from the Italian
Registry.
Am J Med.
1996;100:530-536.
MEDLINE
4.
Bick RL.
Antiphospholipid thrombosis syndromes: etiology, pathophysiology, diagnosis and
management.
Int J Hematol.
1997;65:193-213.
MEDLINE
5.
Bick RL.
The antiphospholipid thrombosis syndromes: fact, fiction, confusion and
controversy.
Am J Clin Pathol.
1993;100:477-480.
MEDLINE
6.
Petri M.
Epidemiology of the antiphospholipid syndrome.
In: Asherson RA, Cervera R, Piette JC, Schoenfeld Y, eds. The Antiphospholipid Syndrome. Boca Raton,
Fla: CRC Press; 1996:13-28.
7.
Bick RL, Jakway J, Baker WF Jr.
Deep vein thrombosis: prevalence of etiologic factors and results of management
in 100 consecutive patients.
Semin Thromb Hemost.
1992;18:267-274.
MEDLINE
8.
Bick RL, Kaplan H.
Syndromes of thrombosis and hypercoagulability.
Med Clin North Am.
1998;82:409-458.
MEDLINE
9.
Moll S, Ortel TL.
Monitoring warfarin therapy in patients with lupus anticoagulants.
Ann Intern Med.
1997;127:177-185.
MEDLINE
10.
DeStefano U, Chirrolo P, Paciaroni K, et al.
Epidemiology of factor V Leiden: clinical implications.
Semin Thromb Hemost.
1998;24:367-379.
MEDLINE
11.
Dahlback B.
New molecular insights into the genetics of thrombophilia: resistance to
activated protein C caused by Arg 506 to Gln mutation in factor V as a
pathogenic risk factor for venous thrombosis.
Thromb Haemost.
1995;74:139-148.
MEDLINE
12.
Chan WP, Lee CK, Kwong YL, et al.
A novel mutation of Arg 306 of factor V gene in Hong Kong Chinese.
Blood.
1998;91:1135-1139.
MEDLINE
13.
Williamson D, Brown K, Luddington R, et al.
Factor V Cambridge: a new mutation (Arg 306 to Thr) associated with resistance
to activated protein C.
Blood.
1998;91:1140-1144.
MEDLINE
14.
Alhenc-Gelas M, Nicaud V, Gandrille S, et al.
The factor V gene A4070G mutation and the risk of venous thrombosis.
Thromb Haemost.
1999;81:193-197.
MEDLINE
15.
Ridker PM, Hennekens CH, Lindpainter K, et al.
Mutation in the gene coding for coagulation factor V and the risk of myocardial
infarction, stroke and venous thrombosis in apparently healthy young men.
N Engl J Med.
1995;332:912-917.
MEDLINE
16.
Siscovick DS, Schwartz SM, Rosendaal FR, et al.
Thrombosis in the young: effect of atherosclerotic risk factors on the risk of
myocardial infarction associated with prothrombotic factors.
Thromb Haemost.
1997;78:7-12.
MEDLINE
17.
Rosendaal FR, Koster T, Vandenbroucke JP, et al.
High risk of thrombosis in patients homozygous for factor V Leiden.
Blood.
1995 Mar 15;85(6):1504-1508.
18.
Martinelli I, Mannucci PM, DeStefano V, et al.
Different risks of thrombosis in four coagulation defects associated with
inherited thrombophilia: a study of 150 families.
Blood.
1998;92:2353-2358.
MEDLINE
19.
Simioni P, Sanson BJ, Prandoni P, et al.
Incidence of venous thromboembolism in families with inherited thrombophilia.
Thromb Haemost.
1999;81:198-202.
MEDLINE
20.
Rodeghiero F, Tosetto A.
Activated protein C resistance and factor V Leiden mutation are independent
risk factors for venous thromboembolism.
Ann Intern Med.
1999;130:643-650.
MEDLINE
21.
Faioni EM, Razzari C, Martinelli I, et al.
Resistance to activated protein C in unselected patients with arterial and
venous thrombosis.
Am J Hematol.
1997;55:59-64.
MEDLINE
22.
Hille ET, Westenderp RG, Vandenbroucke JP, et al.
Mortality and causes of death in families with the factor V Leiden mutation
(resistance to activated protein C).
Blood.
1997;89:1963-1967.
MEDLINE
23.
van der Meer FJM, Koster T, Vandenbroucke JP, et al.
The Leiden Thrombophilia Study.
Thromb Haemost.
1997;78:631-635.
MEDLINE
24.
Koster T, Blann AD, Briet E, et al.
Role of clotting factor VIII in effect of von Willebrand factor on occurrence
of deep vein thrombosis.
Lancet.
1995;345:152-155.
MEDLINE
25.
Kraaijenhagen R, Pieternella S, Koopman M, et al.
High plasma concentrations of factor VIII:C is a major risk factor for venous
thromboembolism.
Thromb Haemost.
2000;83:5-9.
MEDLINE
26.
O'Donnell J, Tuddenham EGD, Manning R, et al.
High prevalence of elevated factor VIII levels in patients referred for
thrombophilia screening: role of increased synthesis and relationship to the
acute phase reaction.
Thromb Haemost.
1997;77:825-828.
MEDLINE
27.
O'Donnell J, Mumford A, Manning R, et al.
Elevation of FVIII:C in venous thromboembolism is persistent and independent of
the acute phase response.
Thromb Haemost.
2000;83:10-13.
MEDLINE
28.
Bloemenkamp KWM, Helmerhorst FM, Rosendaal FR, Vandenbroucke JP.
Venous thrombosis, oral contraceptives and high factor VIII levels.
Thromb Haemost.
1999;82:1024-1027.
MEDLINE
29.
Luzzato G, Schafer AI.
The prethrombotic state in cancer.
Semin Oncol.
1990;17:147-159.
MEDLINE
30.
Rickles FR, Levine M, Edwards RL.
Hemostatic alterations in cancer patients.
Cancer Metastasis Rev.
1992;11:237-248.
MEDLINE
31.
Sun NC, McAfee WM, Hum GJ, et al.
Hemostatic abnormalities in malignancy, a prospective study in one hundred
eight patients, part I: coagulation study.
Am J Clin Pathol.
1979;71:10-16.
MEDLINE
32.
Sack GH, Levin J, Bell W.
Trousseau's syndrome and other manifestations of chronic disseminated
coagulopathy in patients with neoplasms: clinical, pathologic, and therapeutic
features.
Medicine (Baltimore).
1977;56:1-37.
MEDLINE
33.
Mammen EF.
Sticky platelet syndrome.
Semin Thromb Hemost.
1999;25:361-365.
MEDLINE
34.
Mammen EF.
Ten years' experience with the "sticky platelet syndrome.".
Clin Appl Thromb Hemost.
1995;1:66-72.
35.
Tait RC, Walker ID, Reitsma PH, et al.
Prevalence of protein C deficiency in the healthy population.
Thromb Haemost.
1995;73:87-93.
MEDLINE
36.
Griffin JH.
Clinical studies on protein C.
Semin Thromb Hemost.
1984;10:162-166.
MEDLINE
37.
Broekmans AW, Veltkamp JJ, Bertina RM.
Congenital protein C deficiency and venous thromboembolism: a study of three
Dutch families.
N Engl J Med.
1983;309:340-344.
MEDLINE
38.
Broekmans AW.
Hereditary protein C deficiency.
Haemostasis.
1985;15(4):233-240.
MEDLINE
39.
Bauer K.
Hypercoagulable states.
In: Hoffman R, Furie B, Cohen H, Benz E Jr, Silberstein L, Shattil S, eds. Hematology: Basic Principles and Practice.
2nd ed. Camden Town, London, England: Churchill Livingstone Inc;
1995:1781-1794.
40.
Baker W Jr, Bick R.
Treatment of hereditary and acquired thrombophilic disorders.
Semin Thromb Hemost.
1999;25:387-406.
MEDLINE
41.
Engesser L, Broekmans AW, Briet E, et al.
Hereditary protein S deficiency: clinical manifestations.
Ann Intern Med.
1987;106:677-682.
MEDLINE
42.
VanCott EM, LaPosata M.
Laboratory evaluation of hypercoagulable states.
Hematol Oncol Clin North Am.
1998;12:1141-1166.
MEDLINE
43.
den Heijer M, Rosendaal FR, Blom HJ, Gerrits WB, Bos GM.
Hyperhomocysteinemia and venous thrombosis: a meta-analysis.
Thromb Haemost.
1998;80:874-877.
MEDLINE
44.
Rees MM, Rodgers GM.
Homocysteinemia: association of a metabolic disorder with vascular disease and
thrombosis.
Thromb Res.
1993;71:337-359.
MEDLINE
45.
Harpel PC.
Homocysteine, atherogenesis and thrombosis.
Fibrinolysis.
1997;11:77-80.
46.
Bellamy MF, McDowell IFW.
Putative mechanisms for vascular damage by homocysteine.
J Inherit Metab Dis.
1997;20:307-315.
MEDLINE
47.
Franken DG, Boers GHJ, Blom HJ, et al.
Treatment of mild hyperhomocysteinemia in vascular disease patients.
Arterioscler Thromb.
1994;14:465-470.
MEDLINE
48.
Tait RC, Walker ID, Perry DJ, et al.
Prevalence of antithrombin deficiency in the healthy population.
Br J Haematol.
1994;87:106-112.
MEDLINE
49.
Thaler E, Lechner K.
Antithrombin III deficiency and thromboembolism.
Clin Haematol.
1981;10:369-390.
MEDLINE
50.
Marciniak E, Farley CH, DeSimone PA.
Familial thrombosis due to antithrombin III deficiency.
Blood.
1974;43:219-231.
MEDLINE
51.
Francis J.
Laboratory investigation of hypercoagulability.
Semin Thromb Hemost.
1998;24:111-126.
MEDLINE
52.
Menache D.
Replacement therapy in patients with hereditary antithrombin III deficiency.
Semin Hematol.
1991;28:31-38.
MEDLINE
53.
Mammen EF.
Plasminogen abnormalities.
Semin Thromb Hemost.
1983;9:50-51.
54.
Kirschsteln W, Simianer S, Dempfle CE.
Impaired fibrinolytic capacity and tissue plasminogen activator release in
patients with restenosis after PTCA.
Thromb Haemost.
1989;62:772-775.
MEDLINE
55.
DeJong E, Porte RJ, Knot EA.
Disturbed fibrinolysis in patients with inflammatory bowel disease.
Gut.
1989;30:188-194.
MEDLINE
56.
Juhan-Ungue I, Roul C, Alessi MC, et al.
Increased plasminogen activator inhibitor activity in non-insulin dependent
diabetic patients–relationship with plasma insulin.
Thromb Haemost.
1989;61:370-373.
MEDLINE
57.
Ebert R.
Index of Variant Human Fibrinogens.
Boca Raton, Fla: CRC Press; 1994.
58.
Goodnough LT, Saito H, Ratnoff OD.
Thrombosis or myocardial infarction in congenital clotting factor abnormalities
and thrombocytopenias: a report of 21 patients and a review of previously
reported cases.
Medicine (Baltimore).
1983;62:248-255.
MEDLINE
59.
Simioni P, Tormene D, Manfrin D, et al.
Prothrombin antigen levels in symptomatic and asymptomatic carriers of the
20210A prothrombin variant.
Br J Haematol.
1998;103:1045-1050.
MEDLINE
60.
Rosendaal FR, Doggen CJ, Zivelin A, et al.
Geographic distribution of the 20210 G to A prothrombin variant.
Thromb Haemost.
1998;79:706-708.
MEDLINE
61.
Ridker PM, Hennekens CH, Miletich JP.
G20210A mutation in prothrombin gene and risk of MI, stroke, and venous
thrombosis in a large cohort of US men.
Circulation.
1999;99:999-1004.
MEDLINE
62.
Brown K, Luddington R, Williamson D, et al.
Risk of venous thromboembolism associated with a G to A transition at position
20210 in the 3'-untranslated region of the prothrombin gene.
Br J Haematol.
1997;98:907-909.
MEDLINE
63.
Hillarp A, Zoller B, Svensson PJ, et al.
The 20210A allele of the prothrombin gene is a common risk factor among Swedish
outpatients with verified deep venous thrombosis.
Thromb Haemost.
1997;78:990-992.
MEDLINE
64.
Ariens R, Alberio G, Moia M, Mannucci P.
Low levels of heparin-releasable tissue factor pathway inhibitor in young patients
with thrombosis.
Thromb Haemost.
1999;81:203-207.
MEDLINE
65.
Doggen CJ, Kunz G, Rosendaal FR, et al.
A mutation in the thrombomodulin gene, 127G to A coding for Ala25Thr, and the
risk of myocardial infarction in men.
Thromb Haemost.
1998;80:743-748.
MEDLINE
66.
Heron E, Lozinguez O, Alhenc-Gelas M, et al.
Hypercoagulable states in primary upper extremity DVT.
Arch Intern Med.
2000;160:382-386.
ABSTRACT
| FULL TEXT
| PDF
| MEDLINE
67.
Kearon C, Gent M, Hirsh J, et al.
A comparison of three months of anticoagulation with extended anticoagulation
for a first episode of idiopathic venous thromboembolism.
N Engl J Med.
1999;340:901-907.
MEDLINE
68.
Bauer KA.
Management of patients with hereditary defects predisposing to thrombosis
including pregnant women.
Thromb Haemost.
1995;74:94-100.
MEDLINE
Edward E.
Rylander, M.D.
Diplomat American
Board of Family Practice.
Diplomat American
Board of Palliative Medicine.