|
|
Prevention
of Perinatal Group B Streptococcal Disease
Revised
Guidelines from CDC
Prepared by The material in this report was prepared by the National
Center for Infectious Diseases, James M. Hughes, M.D., Director; Division of
Bacterial and Mycotic Diseases, Mitchell L. Cohen, M.D., Director. Summary Group B streptococcus (GBS) remains a
leading cause of serious neonatal infection despite great progress in
perinatal GBS disease prevention in the 1990s. In 1996, CDC, in collaboration
with other agencies, published guidelines for the prevention of perinatal
group B streptococcal disease (CDC. Prevention of perinatal group B streptococcal
disease: a public health perspective. MMWR 1996;45[RR-7]:1--24). Data collected after the issuance of the 1996
guidelines prompted reevaluation of prevention strategies at a meeting of
clinical and public health representatives in November 2001. This report
replaces CDC's 1996 guidelines. The recommendations are based on available
evidence and expert opinion where sufficient evidence was lacking. Although
many of the recommendations in the 2002 guidelines are the same as those in
1996, they include some key changes:
Although universal screening for GBS
colonization is anticipated to result in further reductions in the burden of
GBS disease, the need to monitor for potential adverse consequences of
intrapartum antibiotic use, such as emergence of bacterial antimicrobial
resistance or increased incidence or severity of non-GBS neonatal pathogens,
continues, and intrapartum antibiotics are still viewed as an interim
strategy until GBS vaccines achieve licensure. Introduction
Group B streptococcus (GBS) emerged as the leading
infectious cause of neonatal morbidity and mortality in the United States in
the 1970s (1--4). Initial case
series reported case-fatality ratios as high as 50%. In the early 1980s,
clinical trials demonstrated that administering antibiotics during labor to
women at risk of transmitting GBS to their newborns could prevent invasive
disease in the first week of life (i.e., early-onset disease) (5). As a result of the collaborative
efforts of clinicians, researchers, professional organizations, parent
advocacy groups, and the public health community in the 1990s, recommendations
for intrapartum prophylaxis to prevent perinatal GBS disease were issued in
1996 by the American College of Obstetricians and Gynecologists (ACOG) (6) and CDC (7), and in 1997 by the American Academy of Pediatrics (8). Those guidelines recommended the use of one of two
prevention methods, a risk-based approach or a culture-based screening
approach. Providers using the risk-based method identify candidates for
intrapartum chemoprophylaxis according to the presence of any of the
following intrapartum risk factors associated with early-onset disease:
delivering at <37 weeks' gestation, having an intrapartum temperature >100.4ºF
(>38.0ºC), or rupture of membranes for >18
hours. The screening- based method recommends screening of all pregnant women
for vaginal and rectal GBS colonization between 35 and 37 weeks' gestation.
Colonized women are then offered intrapartum antibiotics at the time of
labor. Under both strategies, women with GBS bacteriuria during their current
pregnancy, or who previously gave birth to an infant with early-onset GBS
disease are candidates for intrapartum antibiotic prophylaxis. Before active prevention was initiated, an estimated
7,500 cases of neonatal GBS disease occurred annually (9). Despite striking declines in disease
incidence coinciding with increased prevention activities in the 1990s, GBS
disease remains a leading infectious cause of morbidity and mortality among
newborns in the United States (10,11).
Moreover, since the release of the 1996 guidelines, new data are available to
evaluate the effectiveness of the screening approach relative to the
risk-based approach and to resolve some of the clinical challenges of
implementing prevention. In light of these new data, in November 2001, CDC
consulted with multiple partners to revise the 1996 guidelines for the
prevention of perinatal group B streptococcal disease, using an
evidence-based approach where possible and scientific opinion when sufficient
data were lacking (Table
1). These updated guidelines replace CDC's 1996 guidelines. They are
intended for the following groups: providers of prenatal, obstetric, and
pediatric care; supporting microbiology laboratories, hospital administrators
and managed care organizations; childbirth educators; public health
authorities; and expectant parents and their advocates. Differences and similarities between
current and previous guidelines
Following are major differences in the new guidelines:
Although important changes have been instituted, many
recommendations remain the same:
Background
Early Infancy and Pregnancy-Related
Infections
GBS causes severe invasive disease in young infants. The
majority of infections in newborns occur within the first week of life and
are designated early-onset disease. Late-onset infections occur in infants
aged >1 week, with most infections evident in the first 3 months of life.
Young infants with invasive GBS disease usually present with sepsis or pneumonia,
and less often contract meningitis, osteomyelitis, or septic arthritis. The
proportion of infants with meningitis is higher among those with late-onset
infections. When neonatal infections caused by GBS appeared in the 1970s, as
many as 50% of patients died. During the 1990s, the case-fatality ratio of
early- and late-onset disease was 4% (10)
because of advances in neonatal care. Intrauterine infection of the fetus results from
ascending spread of GBS from the vagina of a colonized woman who is typically
asymptomatic. Fetal aspiration of infected amniotic fluid can lead to
stillbirth, neonatal pneumonia, or sepsis. Infants can also become infected
with GBS during passage through the birth canal, although the majority of
infants who are exposed to the organism through this route become colonized
on skin or mucous membranes but remain asymptomatic. In pregnant women, GBS can cause clinical infections, but
most women have no symptoms associated with genital tract colonization.
Urinary tract infections caused by GBS complicate 2%--4% of pregnancies (12,13). During pregnancy or the
postpartum period, women can contract amnionitis, endometritis, sepsis, or
rarely, meningitis caused by GBS (14--19).
Fatalities among women with pregnancy-associated GBS disease are extremely
rare. GBS Colonization
The gastrointestinal tract serves as the natural
reservoir for GBS and is the likely source of vaginal colonization. Vaginal
colonization is unusual in childhood but becomes more common in late
adolescence (20). Approximately
10% to 30% of pregnant women are colonized with GBS in the vagina or rectum (21). GBS colonization can be transient,
chronic, or intermittent. Maternal intrapartum GBS colonization is a major
risk factor for early-onset disease in infants, and vertical transmission of
GBS from mother to fetus primarily occurs after the onset of labor or
membrane rupture. However, colonization early in pregnancy is not predictive
of neonatal sepsis (22).
Culture screening of both the vagina and rectum for GBS late in gestation
during prenatal care can detect women who are likely to be colonized with GBS
at the time of delivery and are thus at higher risk of perinatal transmission
of the organism (23). Classic epidemiologic studies conducted during the 1980s
revealed that women with prenatal GBS colonization were >25 times more
likely than women with negative prenatal cultures to deliver infants with
early-onset GBS disease (24).
Researchers used prenatal cultures as the basis for identifying candidates
for intrapartum antimicrobial chemoprophylaxis; clinical trials identified
reductions in vertical transmission of the organism, as measured by infant
colonization (25,26) or by
protection against early-onset disease (5,27).
Heavy colonization, defined as culture of GBS from direct plating rather than
only from selective broth, is associated with higher risk for early-onset
disease. GBS identified in clean-catch urine specimens is considered a
surrogate for heavy maternal colonization and also is associated with a higher
risk for early-onset GBS disease (12,13);
it has been included among indications for intrapartum antibiotic
prophylaxis. GBS Culture-Based Screening Methods
Numerous studies have documented that the accuracy of
prenatal screening cultures in identifying intrapartum colonization status
can be enhanced by careful attention to the timing of cultures, the anatomic
sites swabbed, and the precise microbiologic methods used for culture and
detection of organisms (Box 1). Collection
of cultures between 35 and 37 weeks' gestation is recommended to improve the
sensitivity and specificity of detection of women who remain colonized at the
time of delivery (23,28).
Swabbing both the lower vagina and rectum (i.e., through the anal sphincter)
increases the yield substantially compared with sampling the cervix or
sampling the vagina without also swabbing the rectum (29). Studies have indicated that when
women in the outpatient clinic setting collect their own screening specimens,
with appropriate instruction, GBS yield is similar to when specimens are
collected by a health-care provider (30).
Although swabbing both sites is recommended and use of two swabs can be
justified, both swabs should be placed in a single broth culture medium
because the site of isolation is not important for clinical management and
laboratory costs can thereby be minimized. Because vaginal and rectal swabs
are likely to yield diverse bacteria, use of selective enrichment broth is recommended
(Box 1) to
maximize the isolation of GBS and avoid overgrowth of other organisms. When
direct agar plating is used instead of selective enrichment broth, as many as
50% of women who are GBS carriers have false-negative culture results (31). Additional Risk Factors for Perinatal
GBS Disease
In addition to colonization with GBS, other factors
increase the risk for early-onset disease. These include gestational age
<37 completed weeks, longer duration of membrane rupture, intraamniotic
infection, young maternal age, black race, Hispanic ethnicity, and low
maternal levels of anticapsular antibody (32--37).
In a 1985 report of predictors of early-onset disease (24), women with gestation <37 weeks,
membrane rupture of >12 hours, or intrapartum temperature >99.5ºF
(37.5ºC) had 6.5 times the risk of having an infant with
early-onset GBS disease compared with women with none of those factors. Of
note, women who had one of these risk factors but who had negative prenatal
screening cultures were at relatively low risk for early-onset GBS disease
(attack rate 0.9 per 1,000 births) compared with women who were colonized
prenatally but had none of the risk factors (attack rate 5.1 per 1,000 births)
(24). In a risk-based strategy
promoted during the 1990s as an alternative to prenatal culture-based
screening approaches, prematurity (gestation <37 weeks), intrapartum fever
(temperature >100.4ºF or 38ºC), or duration
of membrane rupture >18 hours were used as clinical indications for
intrapartum prophylaxis. Previous delivery of an infant with invasive GBS
disease may increase the risk of early-onset disease in subsequent deliveries
(38,39), and intrapartum
treatment of such women in subsequent pregnancies has been promoted. By
contrast, colonization with GBS in a previous pregnancy is not considered an
indication for intrapartum prophylaxis in subsequent pregnancies; rather,
women require evaluation for prenatal colonization in each pregnancy. Because
colonization is transient, the predictive value of culture-based screening is
too low to be clinically useful when performed more than 5 weeks before
delivery (28); thus, many women
with GBS colonization during one pregnancy will no longer be colonized during
subsequent pregnancies. Impact
and Implementation of the 1996 Guidelines
Declines in Perinatal GBS Disease
Incidence in the Era of Chemoprophylaxis
Before the widespread use of intrapartum antibiotics, the
incidence of invasive neonatal GBS disease ranged from 2 to 3 cases per 1,000
live births (9,40). Active,
population-based surveillance in selected states in 1990, when GBS prevention
was still rarely implemented, projected an incidence of 1.8 cases per 1,000
live births in the United States (early-onset disease: 1.5/1,000; late-onset:
0.35/1,000) (9). Coinciding with active prevention efforts in the 1990s,
the incidence of early-onset disease declined by 70% to 0.5 cases per 1,000
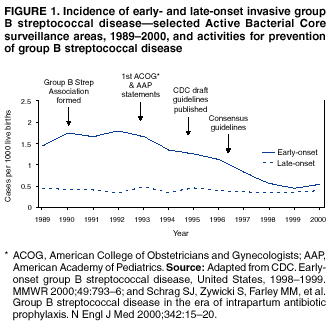
live births in 1999 (Figure
1). Projections from active surveillance data for 1999 from the Active
Bacterial Core surveillance/Emerging Infections Program Network (ABCs)(41) estimate that intrapartum
antibiotics prevented nearly 4,500 early-onset cases and 225 deaths that year
(10,11). Other countries that
have adopted perinatal GBS disease prevention guidelines similar to the
United States have seen comparable declines in early-onset disease incidence
(42--44). Recent estimates of
early-onset disease incidence in the United States suggest a slight increase
in incidence from 1999 to 2000, consistent with a plateau in the impact of
prevention efforts (Figure
1). The incidence of invasive GBS infections among pregnant
women in the United States declined by 21% from 0.29 per 1,000 live births in
1993 to 0.23 in 1998 (10),
suggesting that increased use of intrapartum antibiotics also prevented some
cases of maternal GBS amnionitis and endometritis. In contrast, the rate of
late-onset disease remained fairly constant throughout the 1990s (Figure 1). Although
intrapartum chemoprophylaxis for women with heavy GBS colonization may
prevent a portion of late-onset disease, the stable incidence of late-onset
disease during a period when use of intrapartum antibiotics was increasing
suggests that this intervention is not effective against late-onset disease. Implementation of Chemoprophylaxis
Strategies After the Release of the 1996 Guidelines
Declines in perinatal GBS disease incidence in the 1990s
suggest that prevention strategies have been implemented successfully.
Several studies have explored directly the challenges of implementation and
extent of compliance with recommendations. Surveys of prenatal care providers
in Connecticut and Minnesota in 1998 found that over 80% had a GBS prevention
policy (Connecticut, 95%; Minnesota, 85%) (45).
In Minnesota, family physicians were less likely to have a policy than were
obstetrician/gynecologists and certified nurse midwives (45). A national survey of ACOG members
in 2000 found that 98% of respondents had a GBS prevention policy; 75% of
respondents reported using a version of the culture-based screening approach
(46). Providers in all three
surveys scored well on questions about their knowledge of the screening and
risk-based strategies (45,46). In hospitals that established or revised policies for GBS
prevention shortly after the release of the 1996 guidelines, rates of
early-onset GBS disease declined by 1997 (47).
By 1999, although only 63% of hospitals in a multistate survey of hospitals
in the ABCs areas had a formal GBS prevention policy (48), having a hospital policy was no
longer associated with changes in incidence of GBS disease, likely because a
high proportion of individual practitioners had adopted policies by this
time. Several studies of single institutions or health
maintenance organizations have evaluated adherence of hospital personnel to
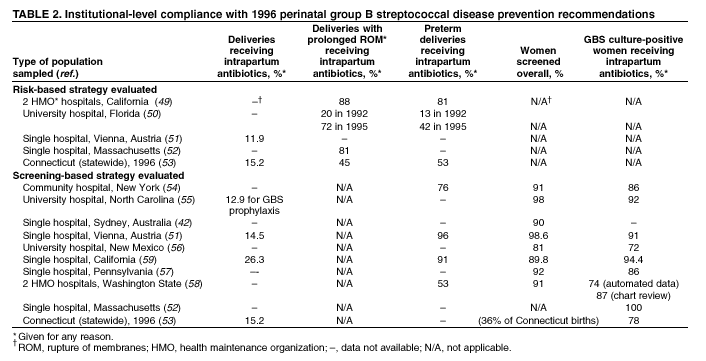
GBS guidelines (Table
2). Among hospitals with a risk-based policy, intrapartum antibiotics
were administered in 40%--80% of preterm deliveries or deliveries with
prolonged rupture of membranes (Table 2) (49--53). Among hospitals with a
culture-based screening policy, close to 90% of delivering women had
documented GBS screening, and close to 90% of GBS-positive women received
intrapartum antibiotics (Table 2) (42,51,54--59). Correct laboratory processing of culture specimens (Box 1) plays a
critical role in successful implementation of the screening policy. A survey
of clinical laboratories in selected counties of three states in 1997--1998
found that only a proportion of laboratories were using the recommended
selective broth media to process GBS cultures (Georgia, 39% of laboratories;
Minnesota, 42%; Connecticut, 62%), suggesting that this may be an area in
need of improvement (31). Although surveys of practitioners and laboratories and
reports from single hospitals help monitor implementation of GBS prevention
guidelines, a recent CDC-sponsored review of labor and delivery records in
selected counties of eight states in the ABCs areas in 1998 and 1999 sheds
light on actual provider practices 2 to 3 years after the release of the 1996
guidelines (60). In this
population, GBS screening was documented in 52% of deliveries, although this
varied widely, from 24% in selected counties of Oregon to 70% in Maryland.
Among screened women, 24% were GBS positive, consistent with carriage rates
reported in earlier studies; 89% of GBS- positive women received intrapartum
antibiotics. The median time of GBS culture collection was at 35.6 weeks' gestation,
consistent with the recommendation of 35--37 weeks' gestation. Among
unscreened women, 24% had at least one intrapartum risk factor; however, only
61% of women with at least one risk factor received intrapartum antibiotics.
Preterm delivery (<37 weeks' gestation) was the most common indication for
which intrapartum antibiotics were not administered. Thus, this multistate
record review confirmed trends in adherence identified in reports from single
hospitals (Table 2).
Maximizing
Prevention by Chemoprophylaxis
Effectiveness of the Risk-Based Approach
Versus the Screening Approach
Despite dramatic declines in GBS incidence in the United
States in the 1990s, GBS remains a leading cause of newborn morbidity and
mortality, resulting in an estimated 1,600 early-onset cases and 80 deaths
annually. Although alternatives to intrapartum antibiotics such as a vaccine
may become available in the future, intrapartum chemoprophylaxis remains the
most effective available intervention against perinatal GBS disease. However,
debate about the most effective strategy for identifying candidates for
intrapartum chemoprophylaxis continues. When the 1996 guidelines were issued, data regarding the
relative effectiveness of the risk-based and screening approaches were not
available. Theoretical predictions based on population estimates of the
proportion of early-onset GBS cases without obstetric risk factors
(approximately 45% in the preprevention era [61])
suggested that the screening-based approach would lead to greater declines in
disease incidence than the risk-based approach (61,62). However, because implementation of the risk-based
approach has been viewed as simpler than the screening-based approach, which
requires correct specimen collection at the prenatal clinic, appropriate
laboratory processing, and timely reporting of results to delivery staff, the
actual effectiveness of these strategies is unknown. Consequently, since
1996, both approaches have been recommended as equally acceptable pending
further data (6--8). Although observational data are now available suggesting
that each strategy can lead to reduced incidence of early-onset GBS disease (49,50,63--65), the strategies have not
been directly compared by clinical trial because of the large sample size
required. A series of single hospital analyses finding benefits of screening
over the risk-based approach (51,56,59,66)
were limited by sequential use of the strategies and inability to control for
potential confounders. A recent CDC-sponsored multistate study provided the
first large-scale direct comparison of the strategies (60). By incorporating population-based
surveillance for early-onset GBS disease into a sample survey of a population
of over 600,000 live births, this analysis found that the screening approach
was >50% more effective than the risk-based approach at preventing
perinatal GBS disease. The protective effect of the screening approach was
robust and persisted after controlling for risk factors associated with
early-onset GBS disease (e.g., preterm delivery, prolonged membrane rupture,
young maternal age, black race). The benefit of screening stemmed from two
main factors. First, by identifying GBS-colonized women who did not present
with obstetric risk factors, screening reached more of the population at risk
than did the risk-based approach. Among the cohort of screened women, 18% of
all deliveries were to mothers who were colonized with GBS but did not have
obstetric risk factors. The efficacy of intrapartum antibiotics in preventing
early-onset GBS disease among infants in this cohort was close to 90%,
suggesting that chemoprophylaxis of GBS-positive women without obstetric risk
factors resulted in significant prevention of early-onset disease. Women who were GBS positive in the screening cohort were
also more likely to receive intrapartum antibiotics than were women with
obstetric risk factors in the risk cohort. Although improvements in
implementation of the risk-based approach would lead to further decline in
disease, this would not be as great as with universal screening (60). Finally, because the effectiveness of screening in this
study was based on actual implementation of this strategy in clinical
practice in 1998 and 1999, further improvements in screening implementation
(e.g., improvements in specimen collection and the methods used for
processing cultures) are expected to result in further benefits. Rationale for a Universal Prenatal
Screening Strategy to Detect GBS Status
The new availability of category II evidence (Table 1) for a
large protective effect of prenatal GBS screening compared with the
risk-based approach provides the foundation for a recommendation of universal
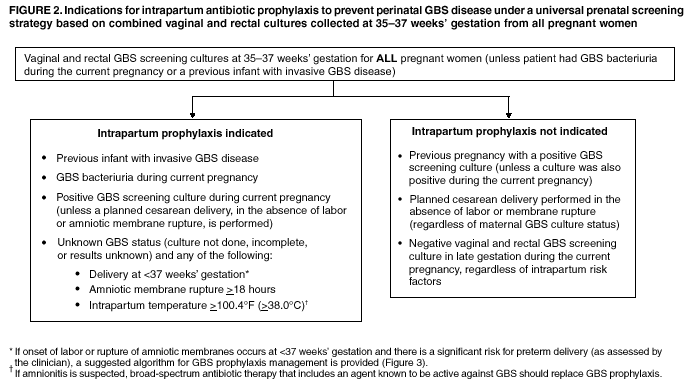
prenatal GBS screening (Figure 2).
Statewide prevention activities in some ABCs areas further demonstrate that
culture-based screening can be successfully implemented in a variety of
settings and institutions. For example, a health department-led survey of
clinical laboratories in Connecticut followed by rapid feedback of survey
results found that the proportion of laboratories in Connecticut using the
correct media for processing GBS screening cultures increased from 62% in
1997 to 92% in 1998 (67) and
100% in 2000. Moreover, coinciding with an active prevention campaign
launched by the state health department that advocated the screening-based
approach, the incidence of early-onset GBS disease in Connecticut declined
from 0.6 cases per 1,000 live births in 1996 (68) to 0.2 cases per 1,000 live births in 1999. From the standpoint of implementation, universal
screening has two additional benefits over the dual recommendations of 1996.
Communication of the public health messages associated with a single strategy
is simpler than communicating and educating about multiple strategies.
Additionally, screening has clear indicators that facilitate evaluation of
implementation (e.g., documentation of GBS test, timing of test, rates of GBS
positivity) (58) compared with
the risk-based approach, in which evidence of prevention implementation
cannot be assessed for approximately 75% of deliveries because they have no
intrapartum risk factors. Cost-effectiveness analyses of the screening- and
risk-based strategies (62,69--73)
have indicated that although the initial costs associated with specimen
collection and processing make the screening strategy more expensive than the
risk-based approach, the overall cost savings due to disease prevention do
not differ importantly between strategies. Additionally, multistate review of
labor and delivery records in 1998 and 1999 suggests that perfect
implementation of the screening- or risk-based strategies will result in a
comparable proportion of deliveries in which women receive intrapartum
antibiotic prophylaxis for GBS (24% for both strategies) (60,74). Thus, the strategies cannot be
distinguished in terms of the proportion of deliveries that will be exposed
to intrapartum antibiotics. Adverse
Effects and Unintended Consequences of Chemoprophylaxis
Potential adverse or unintended effects of GBS prevention
efforts that have raised concern include allergic or anaphylactic reactions
to agents used for intrapartum antibiotic prophylaxis, emergence of GBS
strains resistant to standard therapies, and increasing incidence of serious
neonatal infections caused by pathogens other than GBS, including
antimicrobial-resistant strains. Because of the increasing emergence of
bacterial resistance to antimicrobial agents in both nosocomial and community
settings, assessment of the impact and continued effectiveness of
interventions based on antimicrobial prophylaxis is critical. Antibiotic Allergies Including
Anaphylaxis
Anaphylaxis associated with GBS chemoprophylaxis occurs
but is sufficiently rare that any morbidity associated with anaphylaxis is
greatly offset by reductions in the incidence of maternal and neonatal
invasive GBS disease. Anaphylaxis-related mortality is likely to be a rare
event since women receiving intrapartum antibiotics will be in hospital
settings where rapid intervention is readily available. Estimates of the rate
of anaphylaxis caused by penicillin range from 4/10,000 to 4/100,000 recipients.
Additionally, as many as 10% of the adult population have less severe
allergic reactions to penicillin (75).
Anaphylaxis associated with GBS prophylaxis was reported in the early 1990s (76); since the release of the 1996
guidelines, an additional report of a nonfatal case of anaphylaxis associated
with GBS chemoprophylaxis has been published (77). In a CDC multistate sample of over 5,000 live births,
a single, nonfatal anaphylactic reaction was noted among the 27% of
deliveries in which intrapartum antibiotics were administered (60). In that case, a single dose of
penicillin was administered approximately 4 hours before a preterm cesarean
delivery, and an anaphylactic reaction occurred shortly after the mother
received a single dose of a cephalosporin following umbilical cord clamping. Resistance in GBS
GBS isolates with confirmed resistance to penicillin or
ampicillin have not been observed to date (78--83).
Penicillin remains the agent of choice for intrapartum antibiotic
prophylaxis. Ampicillin is an acceptable alternative, but penicillin is
preferred because it has a narrower spectrum of antimicrobial activity and
may be less likely to select for resistant organisms. The efficacy of both
penicillin (27) and ampicillin
(5) as intrapartum agents for
the prevention of early-onset neonatal GBS disease has been demonstrated in
clinical trials. Although the intramuscular route of administration for
penicillin has been evaluated (25),
intravenous administration is the only route of administration recommended
for intra-partum chemoprophylaxis to prevent perinatal GBS disease,
regardless of the antimicrobial agent used, because of the higher
intraamniotic concentrations achieved with this method. In contrast, the proportions of GBS isolates with in
vitro resistance to clindamycin and erythromycin have increased since 1996.
The prevalence of resistance among invasive GBS isolates in the United States
and Canada ranged from 7% to 25% for erythromycin and from 3% to 15% for
clindamycin in reports published between 1998 and 2001(79--81,84). Resistance to erythromycin
is frequently but not always associated with clindamycin resistance.
Resistance of GBS isolates to cefoxitin, a second-generation cephalosporin
sometimes used as a component of broad-spectrum coverage for
chorioamnionitis, has also been reported (85);
cefoxitin resistance has similarly been observed among invasive GBS isolates
collected from 1996 to 2000 as part of CDC's active surveillance. Whether in
vitro resistance of GBS has direct clinical implications remains unclear (86). Despite emerging resistance to some
drug classes, minimum inhibitory concentrations of cefazolin, a
first-generation cephalosporin available in an intravenous formulation, were
low (<0.5 µg/ml) among a sample of invasive U.S. isolates from 1996
to 2000 (87), suggesting that
GBS isolates are currently susceptible to this agent. Although NCCLS
guidelines do not specify susceptibility breakpoints for cefazolin, they
recommend that all isolates susceptible to penicillin be considered
susceptible to cefazolin (88). In light of the increasing prevalence of resistance to
clindamycin, erythromycin, or both, recommended strategies for providing
intrapartum antibiotic prophylaxis to penicillin-allergic women are updated (Box 2). Because the
efficacy of recommended alternatives to penicillin or ampicillin has not been
measured in controlled trials, and because some of the recommended
alternatives have a broad spectrum of activity and may be more complicated
and costly to administer, verification of a reported history of penicillin
allergy is important. Patients with reported penicillin allergy should then
be assessed to determine their risk for anaphylaxis. Persons at high risk for
anaphylaxis are those who have had immediate hypersensitivity reactions to
penicillin (e.g., anaphylaxis, angioedema, or urticaria) or who have a
history of asthma or other conditions that would make anaphylaxis more
dangerous (89,90). An estimated
10% of persons with penicillin allergy also have immediate hypersensitivity
reactions to cephalosporins (90).
Among penicillin-allergic women not at high risk for anaphylaxis, cefazolin,
because of its narrow spectrum of activity and ability to achieve high
intraamniotic concentrations, is the agent of choice for intrapartum
chemoprophylaxis. For penicillin-allergic women at high risk for
anaphylaxis, testing of GBS isolates from prenatal screening for susceptibility
to clindamycin and erythromycin is recommended if feasible (Box 1). One of
these agents should be employed for intrapartum GBS prophylaxis if the
screening isolate is susceptible to both agents. Vancomycin should be reserved for penicillin-allergic
women at high risk for beta-lactam anaphylaxis when clindamycin or
erythromycin are not options because of in vitro resistance or unknown
susceptibility of a prenatal isolate. Vancomycin use is generally restricted
because of emerging vancomycin resistance among some gram-positive organisms
(e.g., vancomycin-resistant enterococcus and vancomycin-resistant Staphylococcus aureus). An estimated
13.8 million hospitalized patients received vancomycin therapy in 1998 (91). If penicillin allergy occurs in
approximately 10% of adults, and 25% of parturients are colonized with GBS
prenatally, approximately 100,000 of the 4 million annual deliveries would
require prophylaxis with vancomycin in the absence of clindamycin and
erythromycin susceptibility testing of GBS prenatal isolates. This represents
a 7% increase in the number of patients exposed to vancomycin. The total
grams of vancomycin used annually would increase by less than 1% if all penicillin-allergic
colonized women received vancomycin prophylaxis. Increased Incidence or Resistance in
Non-GBS Pathogens
Decreases in the incidence of early-onset GBS sepsis have
not usually been accompanied by increases in incidence of early-onset sepsis
caused by other pathogens, including those that are antibiotic resistant.
Most studies, including population-based multicenter studies, have found
stable (59,92,93) or decreasing
(43) rates of non-GBS
early-onset sepsis during a period of increasing use of intrapartum
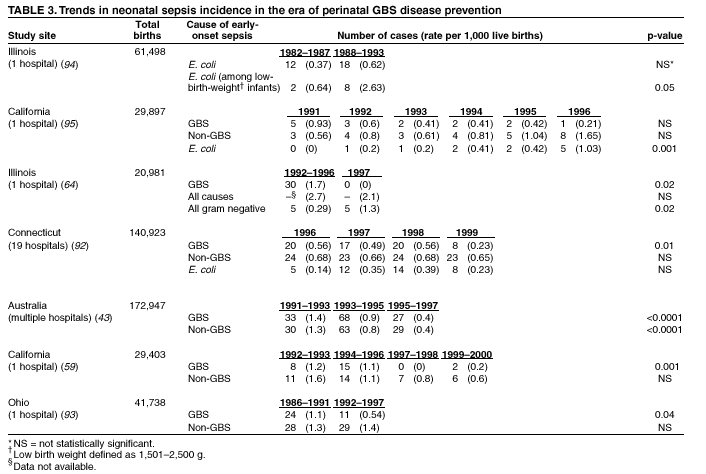
antibiotic prophylaxis for GBS (Table 3). This is
true both for overall non-GBS sepsis and for neonatal sepsis caused by Escherichia coli, the second leading
bacterial cause of neonatal sepsis after GBS (93,94). Some single hospital studies have found increased
rates or case counts of neonatal sepsis caused by E. coli, gram-negative organisms in general, or
ampicillin-resistant pathogens (64,94,95),
but these increases appear to be limited to preterm or low-birth-weight
infants. An increasing proportion of E.
coli neonatal sepsis cases caused by ampicillin-resistant
organisms was observed in two studies (92,94),
but again was limited to preterm or low-birth-weight infants. Furthermore,
the proportion of community-acquired E.
coli infections that are ampicillin resistant has been increasing
(96), suggesting that trends in
antimicrobial resistance should not be attributed to GBS prophylaxis. An association between intrapartum antibiotic exposure
and ampicillin resistance in cases of E.
coli or other non-GBS early-onset sepsis has been observed in
several studies (36,94,95, 97,98).
These reports established that infections caused by antibiotic-resistant
organisms were more frequently preceded by antibiotic use than were
infections caused by susceptible organisms, and that more doses or longer
duration of anti-biotics before delivery increased the chance that a neonatal
infection, if it occurred, would be caused by an antibiotic-resistant
organism. These studies, however, were not designed to assess whether
intrapartum antibiotic use increased the rate of antibiotic-resistant
infections. Moreover, findings from these studies are consistent with
intrapartum antibiotics inducing resistance among initially susceptible
organisms, but also with intrapartum antibiotics preventing
antibiotic-susceptible infections and having no impact on
antibiotic-resistant infections, resulting in a net decrease in the total
rate of infection. The reported increases in antibiotic-resistant
early-onset infections in a few studies are not of sufficient magnitude to
outweigh the benefits of intrapartum antibiotic prophylaxis to prevent
perinatal GBS disease. However, to assure early detection of increases in the
rate of disease or deaths caused by organisms other than GBS, continued
surveillance of neonatal sepsis caused by organisms other than GBS is needed.
Clinical
Challenges
GBS Bacteriuria During Pregnancy
The presence of GBS bacteriuria in any concentration in a
pregnant woman is a marker for heavy genital tract colonization. Therefore,
women with any quantity of GBS bacteriuria during pregnancy should receive
intrapartum chemoprophylaxis. Vaginal and rectal screening at 35--37 weeks is
not necessary for these women. GBS can cause both symptomatic and
asymptomatic urinary tract infections, which should be diagnosed and treated
according to current standards of care for urinary tract infections in
pregnancy. Women with GBS urinary tract infections during pregnancy should
receive appropriate treatment at the time of diagnosis as well as intrapartum
GBS prophylaxis. Laboratory personnel should report any presence of GBS
bacteriuria in specimens obtained from pregnant women. For this to occur,
labeling of urine specimens to indicate that they were obtained from a
pregnant woman is imperative. Planned Cesarean Delivery
Because GBS can cross intact amniotic membranes, a
cesarean delivery does not prevent mother-to-child transmission of GBS.
Moreover, because cesarean delivery itself is associated with health risks
for mother and newborn, GBS colonization of the mother is not an indication
for cesarean delivery, and cesarean delivery should not be used as an alternative
to intrapartum antibiotic prophylaxis for GBS prevention. However, although a risk does exist for transmission of
GBS from a colonized mother to her infant during a planned cesarean delivery
performed before onset of labor in a woman with intact amniotic membranes, it
is extremely low, based on a retrospective study at a single hospital (99) and a review of CDC active,
population-based surveillance data from the 1990s. Thus, in this specific
circumstance, in which the risk for disease is extremely low, the individual
risks to a mother and her infant from receiving intrapartum antibiotic
prophylaxis may balance or outweigh the benefits. Intrapartum antibiotic
prophylaxis to prevent perinatal GBS disease is, therefore, not recommended
as a routine practice for women undergoing planned cesarean deliveries in the
absence of labor or amniotic membrane rupture, regardless of the GBS
colonization status of the mother. Patients expected to undergo planned
cesarean deliveries should nonetheless still undergo routine vaginal and
rectal screening for GBS at 35--37 weeks because onset of labor or rupture of
membranes may occur before the planned cesarean delivery. In rare situations
in which patients or providers opt for intrapartum prophylaxis before planned
cesarean deliveries, administration of antibiotics at the time of incision
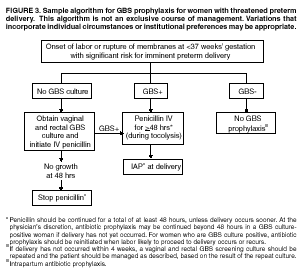
rather than at least 4 hours before delivery may be reasonable (100). Threatened Preterm Delivery
Because preterm (at <37 weeks' gestation) delivery is
an important risk factor for early-onset GBS disease, and because timing of
delivery can be difficult to assess, management of intrapartum prophylaxis
for women with threatened preterm delivery can be challenging. Assessing the
need for intrapartum prophylaxis for these women can also be difficult
because GBS screening is recommended at 35 to 37 weeks' gestation, and
culture results are not always available when labor or rupture of membranes
occur preterm. A suggested approach to GBS chemoprophylaxis in the
context of threatened preterm delivery is outlined (Figure 3). Because
insufficient data are available to suggest a single course of management,
other management strategies developed by individual physicians or institutions
may be appropriate alternatives. The algorithm suggests that if GBS screening
culture results from the current pregnancy are not available and if onset of
labor or rupture of membranes occurs before 37 weeks' gestation with a
substantial risk for preterm delivery (as assessed by the woman's health-care
provider), intrapartum antibiotic prophylaxis for GBS should be provided
pending culture results. For women not yet screened for GBS, a vaginal and
rectal specimen for GBS culture should be obtained if time permits. If a
negative culture result within the previous 4 weeks is on record, or if the
clinician determines that labor can be successfully arrested and preterm
delivery averted, antibiotics for GBS prophylaxis should not be initiated.
Because recent clinical trials suggest that antibiotics administered during
pregnancy may be associated with adverse neonatal outcomes, such as
necrotizing enterocolitis or increased need for supplementary oxygen, without
evident benefit for preterm labor or preterm premature rupture of membranes (101,102), antibiotics should be reserved
for instances in which a significant risk for preterm delivery is present. No data are available on which to recommend a specific
duration of antibiotic administration for GBS-positive women with threatened
preterm delivery when delivery is successfully postponed. Management
strategies based on scientific opinion have been proposed (100); without further data, the
management approach is left to the discretion of the individual provider. Regardless
of management strategy chosen, these women should also receive intrapartum
antibiotic chemoprophylaxis for GBS when labor likely to proceed to delivery
occurs or recurs. Previous data (28)
suggest that the accuracy of GBS screening cultures in predicting
colonization status at delivery is greatest if the cultures are collected
within 5 weeks of delivery. Therefore, if a woman is screened early for GBS
because of threatened preterm delivery but does not deliver within 4 weeks,
she should be screened again for GBS colonization and managed according to
the result of the repeated screening culture (Figure 3). Obstetric Procedures for GBS-Colonized
Women
Questions have arisen regarding whether certain obstetric
procedures, such as digital vaginal examinations, intrauterine fetal
monitoring, and membrane stripping or sweeping to hasten the onset of labor,
should be performed on GBS-colonized women. Asymptomatic GBS colonization is
not an indication to perform any of these procedures. When such procedures
are indicated for other reasons, evidence is currently not sufficient to
recommend that particular procedures should be avoided because of increased
risk of peripartum or perinatal infection. Although some obstetric procedures
(frequent vaginal examinations after onset of labor or membrane rupture [17,36,103--105], intrauterine fetal
monitoring [104,106,107], and
mechanical cervical ripening devices [108])
have been significantly associated with peripartum or perinatal infectious
outcomes, most studies to date have been limited by an inability to randomly
allocate women to treatment groups and have yielded conflicting results.
Moreover, because many studies were performed before GBS prevention was
widely implemented, GBS colonization status was often not known and
intrapartum chemoprophylaxis was less common. A meta-analysis of available
studies examining the use of membrane stripping among women of undetermined
GBS colonization status (109)
found no significant increases in overall peripartum or perinatal infection
rates among women who underwent this procedure and their infants compared
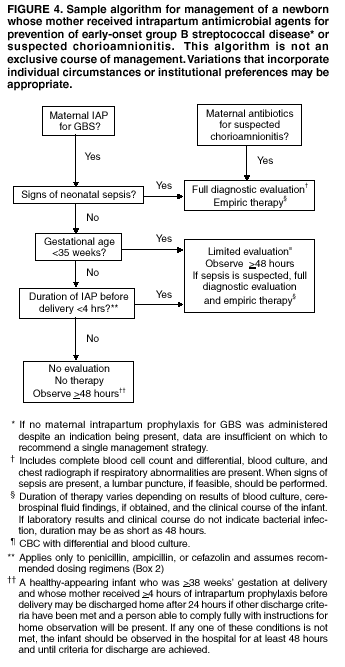
with those who did not. Management of Newborns Exposed to
Intrapartum Prophylaxis
On the basis of information available since the
publication of the 1996 guidelines, a modified approach for empiric
management of newborns born to women who receive intrapartum antibiotics to
prevent early-onset GBS disease or to treat suspected chorioamnionitis is
provided (Figure 4).
Variations in the algorithm that incorporate individual circumstances or
institutional preferences may be appropriate. The modified approach contains
the following changes:
Investigations since 1996 lend additional support to
several components of the algorithm. A retrospective study of over 250,000
live births (115) found that
administration of intrapartum antibiotic prophylaxis did not change the
clinical spectrum of neonatal illness or delay the onset of clinical signs
among infants who contracted GBS disease despite prophylaxis. Thus, the
algorithm targets infants born to mothers with suspected chorioamnionitis and
infants with signs of sepsis for full diagnostic evaluation and empiric
therapy. Also, new evidence indicates that 4 or more hours of intrapartum
ampicillin or penicillin administered according to recommended dosing
intervals (Box 2)
significantly reduces vertical transmission of GBS (116) and risk of early-onset GBS disease
(65). Thus, although the
American Academy of Pediatrics 1997 guidelines suggested 2 or more doses as a
threshold for prophylaxis adequacy for infants >35 weeks' gestation
(8), the revised algorithm
continues to use >4 hours, administered according to recommended
dosing intervals, as the benchmark for optimal prevention of early-onset GBS
disease. Moreover, a review of pregnancies at a West Coast health maintenance
organization using the GBS culture-based screening strategy found that among
women who received intrapartum antibiotic prophylaxis, 50% received
prophylaxis at least 4 hours before delivery, whereas only 14% received at
least 2 doses of intrapartum antibiotics (58);
this indicates that duration of prophylaxis is a more practical target than
number of doses, in addition to being associated with efficacy. One objective of developing an algorithm for management
of newborns was to minimize unnecessary evaluation and antimicrobial
treatment of infants whose mothers received intrapartum prophylaxis. Although
early provider surveys indicated that pediatricians and neonatologists were
more likely to conduct diagnostic evaluations and initiate empiric
anti-biotics for an infant whose mother received intrapartum antibiotic
prophylaxis (117--119), more
recent data indicate that implementation of GBS prevention strategies has not
resulted in increased use of health services for neonates (120), and in some circumstances, when
GBS prophylaxis increased a decrease occurred in the proportion of neonates
who received laboratory evaluations (58).
Intrapartum antibiotic prophylaxis is the method of
choice for preventing neonatal early-onset GBS disease. In the event that
intrapartum antibiotics are not given despite an indication (e.g., delivery
occurred precipitously before antibiotics could be administered to a
GBS-positive woman), sufficient data are not available on which to recommend
a single management strategy for the newborn. Some centers provide
intramuscular penicillin to asymptomatic infants within 1 hour of birth,
based on results of observational studies showing declines in early-onset GBS
disease coincident with a policy of universal administration of intramuscular
penicillin to all newborns (121).
Future
Prevention Technology
Rapid Tests to Detect GBS Colonization
Status
Rapid tests for detection of GBS colonization at the time
of onset of labor or rupture of amniotic membranes might obviate the need for
prenatal culture-based screening if their sensitivity and specificity are
comparable to culture in selective broth media and they yield results rapidly
enough to permit administration of adequate intrapartum antibiotic
prophylaxis to women detected as carriers. Currently available rapid tests
detect GBS antigen from swab specimens. These tests are insufficiently
sensitive to detect light colonization, and therefore are not adequate to
replace culture-based prenatal screening (122,123)
or to use in place of the risk-based approach when culture results are
unknown at the time of labor. An adequate rapid intrapartum test must be as
sensitive as culture (minimally 85% compared with culture of vaginal and
rectal swabs inoculated into selective broth media), rapid so that results
are available to clinicians in time for antibiotics to be given before
delivery, and convenient for integration into routine laboratory use. Even a
highly sensitive rapid detection test would not be adequate if results were not
available to clinicians 24 hours a day, 7 days a week. Alternatives to
culturing vaginal and rectal swab specimens at 35--37 weeks' gestation using
recommended procedures should be validated to show sensitivity similar to
recommended culture methods. A rapid intrapartum test possessing the attributes
described above offers the advantage of ascertaining GBS colonization status
before delivery among women who have had no pre-natal care. Although such
tests might initially be introduced selectively in certain facilities with
sufficient demand and capability, a general recommendation for their use
would require the capacity for effective implementation in a wide range of
hospital settings. Drawbacks of rapid tests include delays in administration
of intrapartum antibiotic prophylaxis while test results are pending and lack
of an isolate for susceptibility testing, which is of particular concern for
penicillin-allergic women. Additionally, until rapid tests are universally
used, missed opportunities for GBS screening may occur among women who
receive prenatal care at institutions relying on intrapartum rapid tests but
who deliver at institutions where such tests are not yet available. In a study of 112 pregnant women at an academic hospital
in Quebec, a new, not yet commercially available fluorogenic polymerase chain
reaction assay was 97% sensitive and 100% specific when compared with vaginal
and rectal cultures collected at admission for delivery. Test results in this
study were available within 45 minutes of specimen collection (124). Further studies are needed to
determine whether this type of test can be adapted for use outside the
research setting. If appropriate techniques for rapid detection of GBS become
commercially available, they may be integrated into the currently recommended
screening strategy. Vaccines To Prevent GBS Disease
Improved use of intrapartum antimicrobial prophylaxis has
resulted in a substantial reduction in early-onset GBS disease, but it is
unlikely to prevent most late-onset neonatal infections, GBS-related
stillbirths, or prematurity, and does not address GBS disease in nonpregnant
adults. Immunization of women during or before pregnancy could prevent
peripartum maternal disease and protect infants from perinatally acquired infection
by transplacental transfer of protective IgG antibodies (125,126). This would eliminate the need
for prenatal GBS screening and intrapartum antimicrobial prophylaxis, along
with associated costs and concerns regarding the potential adverse effects of
intrapartum antibiotic use discussed previously. Serotype-specific antibodies to GBS capsular
polysaccharide, although rare in populations of unvaccinated women, have been
shown to protect against disease (32,127).
Phase 1 and 2 clinical trials among healthy, nonpregnant adults of monovalent
protein-conjugate vaccines containing capsular polysaccharide antigens of GBS
disease-associated serotypes have shown these vaccines to be well tolerated
and immunogenic (128--130). One
challenge of demonstrating vaccine efficacy in preventing early-onset GBS
disease is that the sample size required for clinical trials may be
prohibitively large. Identification of surrogate immunologic measures of
clinical efficacy may thus be important (131,132).
Surrogate information on clinical vaccine efficacy may also be gained by
measuring the impact of multivalent conjugate vaccines on vaginal GBS
colonization (132,133). Anticipated difficulties in making vaccine available to
pregnant women have resulted in consideration of other target populations for
vaccine administration, including adolescent girls (134), women of childbearing age, and
infants (135). The duration of
protection that could be afforded by vaccination is unknown; one or more
booster doses might be required, potentially complicating vaccine delivery.
Shifts in the GBS serotypes causing disease have provided an additional
challenge to vaccine development (133)
and may necessitate modification of vaccine serotype composition over time. Research
Priorities and Tools To Aid Prevention
Technological advances that aid the implementation of a
universal screening strategy will further prevention efforts. In addition to
development of reliable rapid tests that can be performed in a wide range of
labor and delivery settings, methods of simplifying prenatal culture
procedures, e.g., the development of media with a reliable color indicator to
signal presence of GBS, might improve accuracy of prenatal culture results
and facilitate prenatal culture processing at clinical laboratories with
limited technical capacity. Media that have been developed for this purpose,
such as Granada (136,137) or
GBS medium (138), should be
further evaluated to determine if sensitivity and specificity are comparable
to recommended methods, which consist of culture in selective broth media
followed by GBS-specific identification. Although universal prenatal GBS culture-based screening
is likely to result in substantial further declines in the incidence of
early-onset disease, intrapartum chemoprophylaxis is not a permanent or
comprehensive strategy for GBS disease prevention. Because vaccines under
development hold promise to prevent a larger portion of the burden of GBS
disease with a simpler and sustainable intervention, further work on GBS
vaccine development and support of phase 3 clinical trials are warranted (139). Until a safe, effective, and economical vaccine achieves
licensure, it will be important to continue to monitor for potential adverse
effects of chemoprophylaxis, with an emphasis on tracking key sentinel events
signaling a need for revision of the guidelines. Such sentinel events include
the emergence of penicillin resistance among GBS, which to date has not been
detected, and an increase in the incidence of disease or deaths due to neonatal
pathogens other than GBS that offsets the burden of early-onset disease
prevented by chemoprophylaxis. Monitoring for the latter will require
long-term surveillance of a large population of term and preterm births (140). Because GBS carriage is common among delivering women in
the United States, continued surveillance for GBS disease and evaluation of
prevention implementation remains important to minimize missed opportunities
for prevention. States are encouraged to monitor incidence of GBS disease, to
promote activities that enhance perinatal GBS disease prevention and
education, and to assess progress toward national objectives for disease
reduction, such as Healthy People 2010, which sets a target of reducing the
incidence of early-onset GBS disease in all racial and ethnic groups to 0.5
cases per 1,000 live births (141).
Practical tools to assist with monitoring for missed opportunities for
perinatal GBS prevention within hospitals have been published (142); additional prevention information
and tools for providers, patients and clinical microbiologists are available
at http://www.cdc.gov/groupbstrep,
http://www.acog.org, http://sales.acog.com, http://www.aap.org, and http://www.health.state.mn.us/divs/dpc/ades/invbact/strepb.htm.
Recommendations
The following updated recommendations for the prevention
of GBS disease are based on critical appraisal of multistate population-based
observational data and several studies from individual institutions that have
been completed since publication of previous CDC (7), ACOG (6),
and AAP (8) recommendations.
They replace previous recommendations from CDC. The strength (indicated by a
letter) and quality (indicated by a roman numeral) of evidence supporting
each recommendation are shown in parentheses, according to the evidence-based
rating system outlined in Table 1. Obstetric-care practitioners, in conjunction with
supporting laboratories and labor and delivery facilities, should adopt the
following strategy for the prevention of perinatal GBS disease based on
prenatal screening for GBS colonization. The risk-based approach is no longer
an acceptable alternative except for circumstances in which screening results
are not available before delivery (AII).
Before full implementation of this strategy can be
expected in all health-care settings, all members of the health-care team
will need to improve protocols for isolation and reporting of GBS culture
results, to improve information management to ensure communication of
screening results, and to educate medical and nursing staff responsible for
prenatal and intrapartum care. Within institutions, such efforts may take
several months. Even with ideal implementation, cases of early-onset GBS
disease will continue to occur. Tools to help promote prevention and educate
parents of infants with early-onset GBS disease are available at http://www.cdc.gov/groupbstrep.
Additional tools available to assist with prevention implementation are
available at http://www.acog.org, http://sales.acog.com, http://www.aap.org and http://www.health.state.mn.us/divs/dpc/ades/invbact/strepb.htm
Multiple copies of educational materials published by CDC are available at
the Public Health Foundation, 1220 L St., NW Suite 350, Washington, DC 20005,
telephone 877-252-1200, or online at http://www.phf.org.
References
Consultants Kathryn Arnold, M.D., Georgia Division of Public Health,
Atlanta, Georgia; Carol Baker, M.D., American Academy of Pediatrics/
Committee on Infectious Diseases, Elk Grove Village, Illinois; Gina Burns,
M.S., Group B Strep Association, Chapel Hill, North Carolina; Richard
Facklam, Ph.D., CDC, Atlanta, Georgia; Monica Farley, M.D., Infectious
Diseases Society of America, Alexandria, Virginia; Theodore G. Ganiats, M.D.,
American Academy of Family Physicians, Leawood, Kansas; Ronald Gibbs, M.D.,
Infectious Diseases Society of Obstetricians and Gynecologists, Washington,
D.C.; Paul Heath, M.D., U.K. GBS Prevention Working Group, London, England;
James H. Jorgensen, Ph.D., Health Science Center at San Antonio, University
of Texas, San Antonio, Texas; William Kanto, M.D., American Academy of
Pediatrics/Committee on Fetus and Newborn, Elk Grove Village, Illinois;
Shelene Keith, Jesse Cause Foundation--Saving the Babies from Group B Strep,
Port Huenme, California; Tekoa King, M.P.H., American College of Nurse
Midwives, Washington, D.C.; Feng Ying Lin, M.D., National Institute of Child
Health and Development, National Institutes of Health, Bethesda, Maryland;
Ruth Lynfield, M.D., Minnesota Department of Health, Minneapolis, Minnesota;
Martin McCaffrey, M.D., Naval Medical Center, San Diego, California; Elliot
Philipson, M.D., Cleveland Clinic, Cleveland, Ohio; Lisa Porter, M.Ed.,
Jacksonville, Florida; Laura Riley, M.D., American College of Obstetricians
and Gynecologists, Washington, D.C.; Donna Russell, M.S., Washington
Department of Health, Renton, Washington; Pam Sims, Pharm. D., Samford
University, Birmingham, Alabama; Carol A. Spiegel, Ph.D., University of
Wisconsin, Madison, Wisconsin; Barbara Stoll, M.D., Emory University School
of Medicine, Atlanta, Georgia; Beth H. Stover, Healthcare Infection Control
Practices Advisory Committee, CDC, Atlanta, Georgia; Cynthia Whitney, M.D.,
CDC, Atlanta, Georgia; Michael K. Yancey, M.D., Tripler Army Medical Center,
Honolulu, Hawaii; Elizabeth Zell, M.Stat., CDC, Atlanta, Georgia
All MMWR HTML versions of articles are
electronic conversions from ASCII text into HTML. This conversion may have
resulted in character translation or format errors in the HTML version. Users
should not rely on this HTML document, but are referred to the electronic PDF
version and/or the original MMWR
paper copy for the official text, figures, and tables. An original paper copy
of this issue can be obtained from the Superintendent of Documents, U.S. Government
Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800.
Contact GPO for current prices. **Questions or messages regarding errors
in formatting should be addressed to [log in to unmask].
Page converted: 8/6/2002 |
|
||
|
MMWR Home | MMWR Search | Help | Contact
Us This
page last reviewed 8/6/2002
Centers for Disease Control and Prevention
|
Edward E.
Rylander, M.D.
Diplomat American
Board of Family Practice.
Diplomat American
Board of Palliative Medicine.