BMJ 2003;326:185
( 25 January )
Papers
Non-invasive positive pressure ventilation to treat
respiratory failure resulting from exacerbations of chronic obstructive
pulmonary disease: Cochrane systematic review and meta-analysis
Josephine V Lightowler
a Department of Respiratory Medicine,
St James's University Hospital, Leeds LS9 7TF, b Academic
Respiratory Medicine, St Bartholomew's School and the Royal London School of
Medicine and Dentistry, London EC1A 7BE, c Department of
Physiological Medicine, St George's Hospital Medical School, University of
London, London SW17 0RE
Correspondence to: Felix S F Ram [log in to unmask]
|
|
Abstract |
|
|
Objectives: To determine the effectiveness of
non-invasive positive pressure ventilation (NPPV) in the management
of respiratory failure secondary to acute exacerbation of chronic
obstructive pulmonary disease.
Design: Systematic review of randomised
controlled trials that compared NPPV and usual medical care with
usual medical care alone in patients admitted to hospital with
respiratory failure resulting from an exacerbation of chronic
obstructive pulmonary disease and with PaCO2 >6 kPa.
Results: The eight studies
included in the review showed that, compared with usual care alone,
NPPV as an adjunct to usual care was associated with a lower
mortality (relative risk 0.41 (95% confidence interval
0.26 to 0.64)), a lower need for intubation (relative risk
0.42 (0.31 to 0.59)), lower likelihood of treatment failure
(relative risk 0.51 (0.38 to 0.67)), and greater improvements at
1 hour in pH (weighted mean difference 0.03 (0.02 to 0.04)),
PaCO2 (weighted mean difference ![]() 0.40 kPa (
0.40 kPa (![]() 0.78 to
0.78 to ![]() 0.03)), and respiratory
rate (weighted mean difference
0.03)), and respiratory
rate (weighted mean difference ![]() 3.08
breaths per minute (
3.08
breaths per minute (![]() 4.26
to
4.26
to ![]() 1.89)). NPPV
resulted in fewer complications associated with treatment (relative
risk 0.32 (0.18 to 0.56)) and shorter duration of stay in
hospital (weighted mean difference
1.89)). NPPV
resulted in fewer complications associated with treatment (relative
risk 0.32 (0.18 to 0.56)) and shorter duration of stay in
hospital (weighted mean difference ![]() 3.24
days (
3.24
days (![]() 4.42 to
4.42 to ![]() 2.06)).
2.06)).
Conclusions: NPPV should be the
first line intervention in addition to usual medical care to manage
respiratory failure secondary to an acute exacerbation of chronic
obstructive pulmonary disease in all suitable patients. NPPV should
be tried early in the course of respiratory failure and before
severe acidosis, to reduce mortality, avoid endotracheal intubation,
and decrease treatment failure.
|
What is already known on this topic A previous meta-analysis showed NPPV to be an effective
intervention, including for acute exacerbations of COPD, but some studies in
this meta-analysis contained mixed groups of patients and were not of good
quality What this study adds NPPV should be considered early in the course of
respiratory failure and before severe acidosis ensues, to avoid the need for
endotracheal intubation and reduce mortality in patients with COPD |
|
|
Introduction |
|
|
Patients with chronic obstructive pulmonary disease (COPD) are
prone to respiratory failure, often resulting in admission to
hospital. Between a fifth and a third of patients admitted with
hypercapnic respiratory failure secondary to acute exacerbation of COPD
will die in hospital, despite mechanical ventilation.1-5
Conventional treatment aims to ensure adequate continuous
oxygenation and to treat the cause of the exacerbation![]() usually achieved through treatment with
bronchodilators, corticosteroids, antibiotics, and controlled
oxygen. Traditionally, patients who do not respond to conventional
treatment are given invasive ventilation. The procedure of tracheal
intubation and assisted ventilation is associated with high
morbidity, and it may be difficult to wean these patients from
ventilation. 6
7
Furthermore, although it is common practice to give intubation and
mechanical ventilation, complications can result from the intubation
process (damage to local tissue) and during the course of ventilation
(pneumonia and sinusitis associated with ventilators), prolonging
stay in intensive care.8-11
usually achieved through treatment with
bronchodilators, corticosteroids, antibiotics, and controlled
oxygen. Traditionally, patients who do not respond to conventional
treatment are given invasive ventilation. The procedure of tracheal
intubation and assisted ventilation is associated with high
morbidity, and it may be difficult to wean these patients from
ventilation. 6
7
Furthermore, although it is common practice to give intubation and
mechanical ventilation, complications can result from the intubation
process (damage to local tissue) and during the course of ventilation
(pneumonia and sinusitis associated with ventilators), prolonging
stay in intensive care.8-11
Non-invasive positive pressure ventilation (NPPV) is an
alternative treatment for patients admitted to hospital with hypercapnic respiratory
failure secondary to acute exacerbation of COPD.12 In
NPPV the patient receives air or a mixture of air and oxygen from a
flow generator through a full facial or nasal mask, and thus
ventilation is enhanced by the unloading of fatigued ventilatory muscles.
Over the last decade NPPV has been increasingly used as an adjunct
treatment in the management of acute exacerbations of COPD, supported
by a number of case series and randomised controlled trials. 2-4 13-15
However, NPPV is not successful in all cases of acute or chronic
respiratory failure in patients with COPD.16 Failure
rates of between 9% and 50% have been reported. 17 18 We
conducted a systematic review of the literature to determine the
effectiveness of NPPV in patients with respiratory failure resulting
from an acute exacerbation of COPD.
|
|
Methods |
|
|
Inclusion and exclusion criteria
Interventions ![]() Trials were considered for inclusion if the intervention
was NPPV, applied through a nasal or face mask, in addition to usual
medical care. Usual medical care could include supplemental oxygen,
antibiotics, bronchodilators, steroids, respiratory stimulants, and
other suitable interventions (for example, diuretics and
methylxanthines) but could not include treatment with NPPV.
Trials were considered for inclusion if the intervention
was NPPV, applied through a nasal or face mask, in addition to usual
medical care. Usual medical care could include supplemental oxygen,
antibiotics, bronchodilators, steroids, respiratory stimulants, and
other suitable interventions (for example, diuretics and
methylxanthines) but could not include treatment with NPPV.
Types of trials and
participants ![]() We considered
randomised controlled clinical trials of any duration. We excluded
trials where patients had a primary diagnosis of pneumonia, weaning
trials, trials whose patients had other underlying pathologies, and
trials where continuous positive airway pressure or endotracheal
intubation preceded recruitment. All patients entered into the
trials had to have an acute exacerbation of COPD and a baseline PaCO2
at admission of >6 kPa.
We considered
randomised controlled clinical trials of any duration. We excluded
trials where patients had a primary diagnosis of pneumonia, weaning
trials, trials whose patients had other underlying pathologies, and
trials where continuous positive airway pressure or endotracheal
intubation preceded recruitment. All patients entered into the
trials had to have an acute exacerbation of COPD and a baseline PaCO2
at admission of >6 kPa.
Identification and selection of trials
We identified trials by searching the Cochrane Airways Group trials
database, as well as other relevant databases (for example, the
Science Citation Index, PubMed, the UK National Research Register),
up to and including June 2002. No language restrictions were
applied in the retrieval of citations.
We assessed the methodological quality of the trials by using the
Cochrane approach to assessment of allocation concealment: all
trials were scored as "adequate concealment" (grade A),
"uncertain" (grade B), or "clearly inadequate
concealment" (grade C).
|
Data abstraction and
analysis
We used standard forms to abstract all data. Whenever possible we
contacted an author of each trial included in the study to verify
the accuracy of the abstracted data and to obtain further
information. Review Manager version 4.1 (Cochrane Collaboration
software) was used to combine data from the trials. Weighted mean
differences (and 95% confidence intervals) were used to pool data in
continuous variables. For dichotomous variables, relative risks (and
95% confidence intervals) were calculated. The number needed to
treat (and 95% confidence interval) was calculated in an internet
based program called Visual Rx (www.nntonline.net).
We used the DerSimonian and Laird method to test for heterogeneity among
pooled estimates; results were considered significant at the
P<0.05 level. Where heterogeneity was present, the fixed effects model
was used to report results; otherwise the random effects model was
used. If there were sufficient numbers of studies for a particular
outcome, and it was heterogeneous, we investigated it on the basis
of study quality, duration of NPPV, type of NPPV, and type of mask
used to administer NPPV. We also planned funnel plots to detect
publication bias.
An intention to treat analysis was used in all studies except one,
which we excluded from sensitivity analyses.16 We
considered it important that studies use an intention to treat
analysis, as there is anecdotal evidence that some patients drop out
or withdraw after randomisation and at the initiation of treatment,
because of the discomfort of NPPV.
|
|
|
Results |
|
|
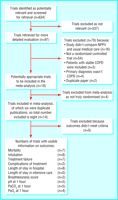
Figure 1
summarises the search for trials and reasons for exclusion, as well as the
numbers of the eight trials included in the review with usable
information on particular outcomes. 2 3 14-17 19 20
Methodological quality of included studies
According to the Cochrane system for grading concealment of
allocation, seven studies were grade A 2 3 14 15 17 19 20 and one
was grade B.16
The seven grade A studies all used the same method for concealing
treatment allocation: a randomly generated sequence of treatment
allocation, contained in sealed envelopes. As all studies were of
good methodological quality, it is unlikely that the quality of the
studies would influence heterogeneity tests or the overall results.
Efficacy variables
We defined treatment failure as the combination of mortality, need
for intubation, and intolerance to the allocated treatment. Data
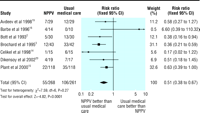
from seven of the studies showed that NPPV resulted in a significantly
lower risk of treatment failure (relative risk 0.51), compared with
usual medical care, with a number needed to treat for NPPV to have a
benefit of five (figure 2,
table 1). 2 3 14-16 19 20 NPPV
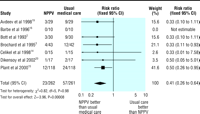
significantly reduced the risk of mortality (relative risk 0.41),
with a number needed to treat of eight (figure 3, table 1). The risk of
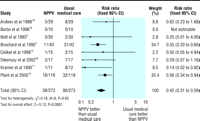
endotracheal intubation was more than halved with NPPV, and for
every five patients treated with NPPV one patient would avoid
intubation (figure 4,
table 1). NPPV also
reduced complications of treatment and length of stay in hospital
(tables 1
and 2). NPPV
significantly improved pH, PaCO2, and respiratory rate within
one hour of initiation (figure 5, table 2).
|
|
|
|
|
|
|
Discussion |
|
|
This systematic review shows a clear benefit of NPPV as an adjunct
treatment to usual medical care in the management of patients admitted
to hospital with respiratory failure secondary to an acute exacerbation
of COPD. NPPV with usual medical care significantly reduces
mortality, endotracheal intubation, treatment failure, complications,
length of hospital stay, and blood gas tensions.
Although NPPV reduces the need for intubation, in some patients
NPPV will fail, and it is essential that a decision be made with the
patient on what should be done in this eventuality. Patients for
whom NPPV eventually fails, despite initial tolerance and effectiveness
of the treatment, need to be distinguished from patients who cannot
tolerate it at all. An uncontrolled study of these "late
failures" suggests a poor outcome regardless of whether the
patient is intubated or continues to receive NPPV.21
NPPV reduced the length of stay in hospital by more than three
days, and length of stay in hospital did not differ between intensive
care units and medical wards. This finding has important resource
implications, given the costs of and pressure on intensive care in
the United Kingdom. However, if NPPV is to be used outside the
intensive care unit (for example, specialist respiratory wards), it
is important that staff are fully trained in the treatment and that
monitoring facilities are in place. It is also important that there
is 24 hour cover by appropriately qualified members of the
medical team.
The number of complications associated with treatment was
significantly lower with NPPV, with an overall risk reduction of 68%.
Almost all of the excess complications occurred because of intubation,
suggesting that avoidance of intubation is the major benefit of
NPPV.
Acidosis is an important prognostic factor for survival after
respiratory failure in COPD, and thus early correction of acidosis is
an essential goal of treatment.5 This
review has shown that NPPV significantly improves pH, PaCO2,
and respiratory rate within the first hour. The improvement in pH
associated with the fall in PaCO2 indicates an
improvement in respiratory failure. A previous study of patients
with respiratory failure secondary to exacerbations of COPD showed
reductions in respiratory rate and transdiaphragmatic activity, with
increases in tidal volume and minute ventilation during NPPV.22 Thus,
NPPV not only improves gas exchange but also facilitates respiratory
muscle rest, reducing the work of respiratory muscles in respiratory
failure, and hence allowing the respiratory muscles to recover and
conventional treatments to work.
Limitations of the review
Publication bias is possible, in that by missing unpublished or
negative trials we may have overestimated the beneficial effect of
NPPV. However, our comprehensive, systematic search strategy of the
literature would minimise any biases. We are confident that most
research in this field was identified. We further minimised bias by
using two independent reviewers, with clearly defined written
inclusion and exclusion criteria for the selection of studies. The
small number of studies meant that a funnel plot analysis for the
detection of publication bias was meaningless. Also, the usefulness
of funnel plots for this purpose is limited by their moderately low
sensitivity.
In none of the studies included in this review was treatment
blinded, because of the practical difficulties of "sham" ventilation.
However, in three of the studies investigators making clinical management
decisions were unaware of which treatment arm a patient was in until
after ventilation began. 2
3 19 In two
studies the decision to intubate was not made by the study
investigators. 14
17 In
another study predefined criteria were used to determine when to
intubate patients in cases of failure of NPPV.15 In one
study there was no indication as to the protocol for intubation and
treatment failure.16
Therefore, we cannot be certain that bias in patient management did
not influence the study outcomes.
The data at one hour would not necessarily include all patients who
were started on treatment, as treatment may have failed (for
example, intubation was necessary or the patient died) before the
one hour time point. Therefore, we may have underestimated the
difference between the two groups in changes in pH, arterial blood
gas tensions, and respiratory rate.
Conclusions
Despite the limitations, this review has shown convincing evidence
from good quality, randomised controlled trials that NPPV is an
effective adjunct to usual medical care in the management of
respiratory failure secondary to acute exacerbations of COPD.
Trialling NPPV should be considered early in the course of
respiratory failure and before severe acidosis ensues, to avoid endotracheal
intubation and treatment failure and to reduce mortality. Further studies
are needed to evaluate the appropriate selection of patients and to
find the best level and schedule of ventilation.
|
|
Acknowledgments |
We thank the members of the Cochrane
Airways Group based at St George's Hospital Medical School, London. We also
thank authors of studies who responded to requests for further data.
2 15 17 19 20
Contributors: FSFR and JAW revised the original review protocol
that was published in 1996 in the Cochrane
Library. FSFR and JVL searched for trials and abstracted and analysed
the data from the included trials. FSFR prepared the manuscript, with input
from JVL, JAW, and MWE. FSFR revised the manuscript for resubmission and is the
guarantor for the paper.
|
|
Footnotes |
Funding: FSFR is funded by the Netherlands Asthma Foundation. JVL
was funded by a British Lung Foundation project grant.
Competing interests: JAW has received educational grant support from
Respironics, one of the manufacturers of nasal ventilators. MWE has
received an honorarium for lecturing from Respironics, has been lent
ventilators for studies from ResMed and Breas, and has had a
contribution from ResMed towards the salary of a research nurse.
![]() A table
showing details of the eight trials included in the systematic review is shown
on bmj.com
A table
showing details of the eight trials included in the systematic review is shown
on bmj.com
|
|
References |
|
|
|
1. |
Ambrosino N, Foglio K, Rubini F, Clini
E, Nava S, Vitacca M. Non-invasive mechanical ventilation in acute
respiratory failure due to chronic obstructive pulmonary disease: correlates
for success. Thorax 1995; 50:
755-757[Abstract].
|
|
2. |
Bott J, Carroll MP, Conway JH, Keilty
SEJ, Ward EM, Brown AM, et al. Randomised controlled trial of nasal ventilation
in acute ventilatory failure due to chronic obstructive airways disease. Lancet 1993; 341: 1555-1557[ISI][Medline].
|
|
3. |
Brochard L, Mancebo J, Wysocki M, Lofaso
F, Conti G, Rauss A, et al. Noninvasive ventilation for acute exacerbations
of chronic obstructive pulmonary disease. N
Engl J Med 1995; 333: 817-822 |
|
4. |
Foglio C, Vitacca M, Quadri A, Scalvini
S, Marangoni S, Ambrosino N. Acute exacerbations in severe COLD patients.
Treatment using positive pressure ventilation by nasal mask. Chest 1992; 101: 1533-1538[Abstract].
|
|
5. |
Jeffrey AA, Warren PM, Flenley DC. Acute
hypercapnic respiratory failure in patients with chronic obstructive lung
disease: risk factors and use of guidelines for management. Thorax 1992; 47: 34-40[Abstract].
|
|
6. |
Brochard L, Rauss A, Benito S, Conti G,
Mancebo J, Rekik N, et al. Comparison of three methods of gradual withdrawal
from ventilatory support during weaning from mechanical ventilation. Am J Respir Crit Care Med 1994; 150:
896-903[Abstract].
|
|
7. |
Esteban A, Frutos F, Tobin MJ, Alia I,
Solsona JF, Valverdu I, et al. A comparison of four methods of weaning patients
from mechanical ventilation. Spanish Lung Failure Collaborative Group. N Engl J Med 1995; 332: 345-350 |
|
8. |
Guerin C, Girard R, Chemorin C, De Varax
R, Fournier G. Facial mask noninvasive mechanical ventilation reduces the
incidence of nosocomial pneumonia. A prospective epidemiological survey from
a single ICU. Intensive Care Med
1997; 23: 1024-1032[ISI][Medline].
|
|
9. |
Fagon JY, Chastre J, Hance A, Montravers
P, Novara A, Gibert C. Nosocomial pneumonia in ventilated patients: a cohort
study evaluating attributable mortality and hospital stay. Am J Med 1993; 94: 281-287[ISI][Medline].
|
|
10. |
Kramer B. Ventilator-associated
pneumonia in critically ill patients. Ann
Intern Med 1999; 130: 1027-1028[ISI][Medline].
|
|
11. |
Nourdine K, Combes P, Carton MJ, Beuret
P, Cannamela A, Ducreux JC. Does noninvasive ventilation reduce the ICU
nosocomial infection risk? A prospective clinical survey. Intensive Care Med 1999; 25: 567-573[ISI][Medline].
|
|
12. |
British Thoracic Society Standards of
Care Committee. Non-invasive ventilation in acute respiratory failure. Thorax 2002; 57: 192-211 |
|
13. |
Meduri GU, Conoscenti CC, Menashe P,
Nair S. Noninvasive face mask ventilation in patients with acute respiratory
failure. Chest 1989; 95: 865-870[Abstract].
|
|
14. |
Celikel T, Sungur M, Ceyhan B, Karakurt
S. Comparison of noninvasive postitive pressure ventilation with standard
medical therapy in hypercapnic acute respiratory failure. Chest 1998; 114: 1636-1642[Abstract].
|
|
15. |
Plant PK, Owen JL, Elliott MW. Early use
of non-invasive ventilation for acute exacerbations of chronic obstructive
pulmonary disease on general respiratory wards: a multicentre randomised
controlled trial. Lancet 2000;
355: 1931-1935[ISI][Medline].
|
|
16. |
Barbe R, Togores B, Rubi M, Pons S,
Maimo A, Agusti AGN. Noninvasive ventilatory support does not facilitate
recovery from acute respiratory failure in chronic obstructive pulmonary
disease. Eur Respir J 1996; 9:
1240-1245[ISI][Medline].
|
|
17. |
Kramer N, Meyer TJ, Meharg J, Cece RD,
Hill NS. Randomized, prospective trial of noninvasive positive pressure ventilation
in acute respiratory failure. Am J Respir
Crit Care Med 1995; 151: 1799-1806[Abstract].
|
|
18. |
Soo Hoo GW, Santiago S, Williams AJ. Nasal
mechanical ventilation for hypercapnic respiratory failure in chronic
obstructive pulmonary disease: determinants of success and failure. Crit Care Med 1994; 22: 1253-1261[ISI][Medline].
|
|
19. |
Avdeev SN, Tretyakov AV, Grigoryants RA,
Kutsenko MA, Chuchalin AG. [Noninvasive positive airway pressure ventilation:
role in treating acute respiratory failure caused by chronic obstructive
pulmonary disease.] Anesteziol Reanimatol
1998; May-Jun: 45-51.
(In Russian.) |
|
20. |
Dikensoy O, Ikidag B, Filiiz A, Bayram
N. Comparison of non-invasive ventilation and standard medical therapy in acute
hypercapnic respiratory failure: a randomised controlled trial at a tertiary
health centre in SE Turkey. Int J Clin
Pract 2002; 56: 85-88[ISI][Medline].
|
|
21. |
Moretti M, Cilione C, Tampieri A,
Fracchia C, Marchioni A, Nava S. Incidence and causes of non-invasive
mechanical ventilation failure after initial success. Thorax 2000; 55: 819-825 |
|
22. |
Brochard L, Isabey D, Piquet J, Amaro P,
Mancebo J, Messadi AA, et al. Reversal of acute exacerbations of chronic
obstructive lung disease by inspiratory assistance with a face mask. N Engl J Med 1990; 323: 1523-1530[Abstract].
|
(Accepted 16 October 2002)
Edward E.
Rylander, M.D.
Diplomat
American Board of Family Practice
Diplomat
American Board of Palliative Medicine