|
|
Advances in Mechanical Ventilation
Martin J. Tobin, M.D. The chief reason that patients are
admitted to an intensive care unit is to receive ventilatory
support. In this article, I update the basic principles of
mechanical ventilation, which I reviewed in the Journal in 1994,1 and
discuss recent advances.
Basic Principles
The indications for mechanical ventilation, as derived from a
study of 1638 patients in eight countries,2 are
acute respiratory failure (66 percent of patients), coma (15
percent), acute exacerbation of chronic obstructive pulmonary
disease (13 percent), and neuromuscular disorders (5 percent). The
disorders in the first group include the acute respiratory distress
syndrome, heart failure, pneumonia, sepsis, complications of
surgery, and trauma (with each subgroup accounting for about 8 to 11
percent of the overall group). The objectives of mechanical
ventilation are primarily to decrease the work of breathing and
reverse life-threatening hypoxemia or acute progressive respiratory
acidosis.
Virtually all patients who receive ventilatory support undergo
assist-control ventilation, intermittent mandatory ventilation, or
pressure-support ventilation; the latter two modes are often used
simultaneously.2
With assist-control ventilation, the most widely used mode, the
ventilator delivers a set tidal volume when triggered by the
patient's inspiratory effort or independently, if such an effort
does not occur within a preselected time.
Intermittent mandatory ventilation was introduced to provide graded
levels of assistance. With this mode, the physician sets the number
of mandatory breaths of fixed volume to be delivered by the
ventilator; between these breaths, the patient can breathe spontaneously.3
Patients often have difficulty adapting to the intermittent nature
of ventilatory assistance, and the decrease in the work of breathing
may be much less than desired.4
Pressure-support ventilation also provides graded assistance but
differs from the other two modes in that the physician sets the
level of pressure (rather than the volume) to augment every spontaneous
respiratory effort.5
The level of pressure delivered by the ventilator is usually
adjusted in accordance with changes in the patient's respiratory
frequency. However, the frequency that signals a satisfactory level
of respiratory-muscle rest has never been well defined, and
recommendations range from 16 to 30 breaths per minute.6
New modes of mechanical ventilation are often introduced. Each
has an acronym, and the jargon is inhibiting to those unfamiliar with
it. Yet each new mode involves nothing more than a modification of
the manner in which positive pressure is delivered to the airway and
of the interplay between mechanical assistance and the patient's
respiratory effort. The purpose of a new mode of ventilation may be
to enhance respiratory-muscle rest, prevent deconditioning, improve
gas exchange, prevent lung damage, enhance the coordination between
ventilatory assistance and the patient's respiratory efforts, and
foster lung healing; the priority given to each goal varies.
Coordinating Respiratory Effort and Mechanical
Ventilation
Probably the most common reason for instituting mechanical
ventilation is to decrease the work of the respiratory muscles. The
inspiratory effort expended by patients with acute respiratory
failure is about four times the normal value, and it can be
increased to six times the normal value in individual patients.7
Critically ill patients in whom this increased level of effort is
sustained indefinitely are at risk of inspiratory-muscle fatigue, which
can add structural injury to already overworked muscles.8 It
is sometimes thought that the simple act of connecting a patient to
a ventilator will decrease respiratory effort. Yet unless the
settings are carefully selected, mechanical ventilation can actually
do the opposite.
With careful selection of ventilator settings, inspiratory effort
can be reduced to the normal range.9 But
eliminating inspiratory effort is not desirable because it causes
deconditioning and atrophy of the respiratory muscles.10
Surprisingly, researchers have not attempted to determine the
desirable target for reducing inspiratory effort in patients with
acute respiratory distress. To reduce effort markedly requires that
the ventilator cycle in unison with the patient's central
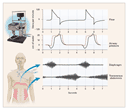
respiratory rhythm (Figure 1). For
perfect synchronization, the period of mechanical inflation must
match the period of neural inspiratory time (the duration of
inspiratory effort), and the period of mechanical inactivity must
match the neural expiratory time.12,13
Difficulties in synchronization can arise at the onset of inspiratory
effort, at the onset of flow delivered by the ventilator, during the
period of ventilator-induced inflation, and at the switch between inspiration
and expiration.
|
Almost all patients who undergo mechanical ventilation receive some
form of assisted ventilation, with the patient's inspiratory effort triggering
the ventilator. To ensure that the ventilator does not cycle too
often, the clinician sets a threshold for airway pressure that will
trigger the ventilator. This threshold, referred to as set
sensitivity, is usually –1 to –2 cm of water.14
To reach this threshold, the patient must initiate an inspiratory
effort. But when the threshold is reached, inspiratory neurons do
not simply switch off. Consequently, the patient may expend
considerable inspiratory effort throughout the machine-cycled inflation.15
The display of airway pressure and flow tracings on ventilator
screens has increased awareness that inspiratory effort is frequently
insufficient to trigger the ventilator. At high levels of mechanical
assistance, up to one third of a patient's inspiratory efforts may
fail to trigger the machine.9,16,17
Surprisingly, unsuccessful triggering is not the result of poor
inspiratory effort; indeed, the effort is more than a third greater
when the threshold for triggering the ventilator is not reached than
when it is reached.9
Breaths that do not reach the threshold for triggering the ventilator
have higher tidal volumes and shorter expiratory times than do
breaths that do trigger the ventilator. Consequently, elastic-recoil pressure
builds up within the thorax in the form of intrinsic positive
end-expiratory pressure (PEEP), or auto-PEEP.9 To
trigger the ventilator, the patient's inspiratory effort first has
to generate a negative intrathoracic pressure in order to
counterbalance the elastic recoil and then must reach the set
sensitivity. The consequences of wasted inspiratory efforts are not
fully known, but they add an unnecessary burden in patients whose
inspiratory muscles are already under stress.
The inspiratory flow rate is initially set at a default value,
such as 60 liters per minute. If the delivered flow does not meet
the patient's ventilatory needs, inspiratory effort will increase.15
Sometimes the flow is increased in order to shorten the inspiratory
time and increase the expiratory time, especially in patients with
inspiratory efforts that are insufficient to trigger the ventilator.
But an increase in flow causes immediate and persistent tachypnea,
and as a result, the expiratory time may be shortened.18
In one study, for example, increases in inspiratory flow from 30
liters per minute to 60 and 90 liters per minute caused increases in
the respiratory rate of 20 and 41 percent, respectively.19
In studies of interactions between the patient's respiratory effort
and mechanical ventilation, remarkably little attention has been
paid to the switch between inspiration and expiration. With the use
of pressure-support ventilation, ventilatory assistance ceases when
the patient's inspiratory flow falls by a preset amount (e.g., to 25
percent of the peak flow).5 Air
flow changes more slowly in patients with chronic obstructive
pulmonary disease than in other patients, and patients often start
to exhale while the ventilator is still pumping gas into their
chests.6,11
In 5 of 12 patients with chronic obstructive pulmonary disease who
were receiving pressure support of 20 cm of water, expiratory muscles
were recruited during ventilator-induced inflation.6
Improving Oxygenation and Preventing Lung Injury
A primary goal of mechanical ventilation is to improve arterial
oxygenation. Improvement is achieved partly through the use of
endotracheal intubation to ensure the delivery of oxygen to the
airway and partly through an increase in airway pressure. Satisfactory
oxygenation is easily achieved in most patients with airway
obstruction. The main challenge arises in patients with
alveolar-filling disorders, especially the acute respiratory distress
syndrome — a form of noncardiogenic pulmonary edema resulting from
severe acute alveolar injury. It has long been recognized that
arterial oxygenation can be achieved at a lower inspired oxygen
concentration by increasing airway pressure. The goal of using the
lowest possible oxygen concentration to achieve an arterial oxygen
saturation of approximately 90 percent has not changed in decades.
What has changed is how this goal is viewed in relation to other
factors, particularly ventilator pressures. In recent years, there
has been a growing tendency to be more concerned about high airway
pressures than about oxygen toxicity, although this shift has been
based on a consensus of opinion rather than on data from studies in
patients and animals.
From the outset, clinicians recognized that mechanical ventilation
could rupture alveoli and cause air leaks.20
In 1974, Webb and Tierney showed that mechanical ventilation could
also cause ultrastructural injury, independently of air leaks.21
Their observations went largely unnoticed until a decade later, when
several investigators confirmed and extended them. Alveolar overdistention
causes changes in epithelial and endothelial permeability, alveolar
hemorrhage, and hyaline-membrane formation in laboratory animals.22
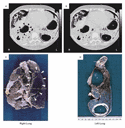
Diffuse infiltrates on chest radiographs originally led clinicians
to infer that lung involvement was homogeneous. But computed tomography
(CT) reveals a patchy pattern: about one third of the lung is
unaerated, one third poorly aerated, and one third normally aerated.23,24 A
ventilator-induced breath will follow the path of least impediment,
travelling preferentially to the normally aerated areas. As a
result, these regions are vulnerable to alveolar overdistention and
the type of ventilator-induced lung injury found in laboratory
animals25
(Figure 2).
|
A new era of ventilatory management began in 1990, when Hickling et
al.26
reported that lowering the tidal volume caused a 60 percent decrease
in the expected mortality rate among patients with the acute
respiratory distress syndrome. In a subsequent trial, Amato et al.27,28
randomly assigned patients to a conventional tidal volume (12 ml per
kilogram of body weight) or to a low tidal volume (less than 6 ml
per kilogram). Mortality was decreased by 46 percent with the lower
tidal volume. In a recent study of 861 patients, the Acute
Respiratory Distress Syndrome Network29
confirmed this benefit: mortality was decreased by 22 percent with
a tidal volume of 6 ml per kilogram as compared with a tidal volume
of 12 ml per kilogram. Lowering the tidal volume, however, failed to
improve the outcome in three controlled trials.30,31,32
The discrepant findings can be explained by differences in trial design.
Increased survival was demonstrable only when the patients undergoing
conventional ventilation had a mean pressure during an
end-inspiratory pause (the so-called plateau pressure, a surrogate
for peak alveolar pressure) that exceeded 32 cm of water.33
The pressures pertinent to ventilatory management are the peak
inspiratory pressure, plateau pressure, and end-expiratory pressure.
Patients with airway obstruction may have a very high peak pressure without
any increase in the plateau pressure. Indeed, the gradient between
the two is directly related to the resistance of the airway to
airflow. An increase in the peak inspiratory pressure without a
concomitant increase in the plateau pressure is unlikely to cause alveolar
damage. The critical variable is not airway pressure itself but
transpulmonary pressure — airway pressure during the end-inspiratory
pause minus pleural pressure. The normal lung is maximally distended
at a transpulmonary pressure between 30 and 35 cm of water, and
higher pressures cause overdistention. Patients with stiff chest
walls, such as those with the acute respiratory distress syndrome
due to a nonpulmonary disorder (e.g., abdominal sepsis), have an
elevated pleural pressure. In such patients, the airway plateau
pressure may exceed 35 cm of water without causing alveolar
overdistention.
Clinical decisions based on plateau pressure must take into account
the relation between lung volume and airway pressure in the
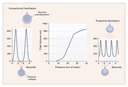
individual patient. The pressure–volume curve in patients with the
acute respiratory distress syndrome typically has a sigmoid shape
with two discrete bends, called inflection points (Figure 3).34
Some investigators believe that a plateau pressure above the upper
bend causes alveolar overdistention. Reducing the tidal volume
lowers the plateau pressure, but at the cost of hypercapnia. In a study
in which 25 patients with the acute respiratory distress syndrome
underwent mechanical ventilation with a tidal volume of 10 ml per
kilogram, 20 had a plateau pressure that was 2 to 14 cm of water
above the upper bend of the pressure–volume curve.35
Lowering the plateau pressure to a value that fell below the upper
bend required a 22 percent decrease in the tidal volume, causing the
partial pressure of carbon dioxide to increase from 44 to 77 mm Hg.35
The partial pressure of carbon dioxide, in turn, can be decreased by
as much as 28 percent by removing tubing and thus decreasing dead
space and increasing the frequency of ventilator-induced breaths. By
virtue of their stiff lungs, patients with the acute respiratory
distress syndrome who do not have an underlying airway obstruction
can tolerate a frequency of 30 breaths per minute without gas
trapping.36
Severe hypercapnia can have adverse effects, including increased
intracranial pressure, depressed myocardial contractility, pulmonary
hypertension, and depressed renal blood flow.37,38
The view that these risks are preferable to the higher plateau
pressure required to achieve normocapnia represents a substantial
shift in ventilatory management.
|
Lowering the tidal volume is not without hazards. In addition to the
potential harm of hypercapnia, the volume of aerated lung may be
decreased,39
with a consequent increase in shunting and worsening oxygenation.
One means of minimizing the loss of lung volume is the use of sighs
(i.e., single breaths of large tidal volume). In one study,
increasing the plateau pressure by at least 10 cm of water during
sighs, applied three times a minute over a period of one hour,
caused a 26 percent decrease in shunting, with a 50 percent increase
in the partial pressure of oxygen.40
It is unknown whether sighs used at this low frequency cause injury
from alveolar overdistention.
The more usual way of improving oxygenation is through the use
of PEEP with the intention of recruiting previously nonfunctioning lung
tissue. Selecting the right level of PEEP for a given patient with
the acute respiratory distress syndrome is difficult, because the
severity of injury varies throughout the lungs. PEEP can recruit
atelectatic areas but may overdistend normally aerated areas.41,42
In a study involving six patients with acute lung injury, for
example, the use of PEEP at 13 cm of water resulted in the
recruitment of nonaerated portions of lung, with a gain of 320 ml in
volume, but three patients had overdistention of already aerated
portions of lung, with an excess volume of 238 ml.43
Overall, about 30 percent of patients with acute lung injury do
not benefit from PEEP or have a fall in the partial pressure of
oxygen.23,44,45
With the patient in the supine posture, PEEP generally recruits the regions
of the lung closest to the apex and sternum.23
Conversely, PEEP can increase the amount of nonaerated tissue in the
regions close to the spine and the diaphragm.23
Among patients in the early stages of the acute respiratory distress
syndrome, those with pulmonary causes, such as pneumonia, are less
likely to benefit from PEEP than are those with nonpulmonary causes,
such as intraabdominal sepsis or extrathoracic trauma.46
This distinction may be related to the type of morphologic involvement:
pulmonary causes of the syndrome are characterized by alveolar filling,
whereas nonpulmonary causes are characterized by interstitial edema
and alveolar collapse. In the later stages of the acute respiratory
distress syndrome, remodeling and fibrosis may eliminate this
distinction between pulmonary and nonpulmonary causes.
To select the right level of PEEP, some experts recommend bedside
calculation of the pressure–volume curve. With the ventilators currently
used in the United States, calculating the pressure–volume curve is
logistically difficult and technically demanding.34
Yet many ventilators have a computer screen, and minor software modifications
would make it feasible to calculate the curve in as little as two
minutes — as with the ventilators available in France.47
Providing this option on ventilators would increase clinicians'
experience with the use of pressure–volume curves in ventilatory management.
Even if the pressure–volume curve is not calculated at the
bedside, it is useful to select the PEEP level according to this
conceptual framework. A level above the lower bend in the
pressure–volume curve is thought to keep alveoli open at the end of
expiration and thus prevent the injury that can result from shear
forces created by the opening and closing of alveoli.48,49,50
This level of PEEP may also prevent an increase in the amount of
nonaerated tissue and, thus, atelectasis. However, the notion that
the lower bend signals the level of PEEP necessary to prevent
end-expiratory collapse and that pressures above the upper bend
signal alveolar overdistention is a gross oversimplification. The
relation between the shape of the pressure–volume curve and events
at the alveolar level is confounded by numerous factors and is the
subject of ongoing research and debate.51,52,53,54,55
An understanding of this relation is also impeded by the difficulty in
distinguishing collapsed lung units from fluid-filled units on CT.
Most patients with the acute respiratory distress syndrome have
an increase in the partial pressure of oxygen when there is a
change from the supine to the prone position. In a study of 16
patients, for example, 12 had an increase of 9 to 73 mm Hg in the
partial pressure of oxygen, and 4 had a decrease of 7 to 16 mm Hg.56
The mechanism responsible for the improvement in the partial
pressure of oxygen is not clear. The attribution of this improvement
to lung recruitment has not been proved.56
It is now posited that a prone position causes ventilation to be
distributed more evenly to the various regions of the lungs,57,58
improving the relation between ventilation and perfusion.59,60
Discontinuing Mechanical Ventilation
Because mechanical ventilation can have life-threatening
complications, it should be discontinued at the earliest possible
time. The process of discontinuing mechanical ventilation, termed
weaning, is one of the most challenging problems in intensive care,
and it accounts for a considerable proportion of the workload of
staff in an intensive care unit.2
When mechanical ventilation is discontinued, up to 25 percent
of patients have respiratory distress severe enough to necessitate the
reinstitution of ventilatory support.61,62
Our understanding of why weaning fails in some patients has advanced
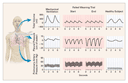
considerably in recent years. Among patients who cannot be weaned,
disconnection from the ventilator is followed almost immediately by
an increase in respiratory frequency and a fall in tidal volume —
that is, rapid, shallow breathing63 (Figure 4). As a
trial of spontaneous breathing is continued over the next 30 to 60
minutes, the respiratory effort increases considerably, reaching
more than four times the normal value at the end of this period.7
The increased effort is mainly due to worsening respiratory mechanics.
Respiratory resistance increases progressively over the course of a
trial of spontaneous breathing, reaching about seven times the
normal value at the end of the trial; lung stiffness also increases,
reaching five times the normal value; and gas trapping, measured as
auto-PEEP, more than doubles over the course of the trial.7 Before
weaning is started, however, the respiratory mechanics in such
patients are similar to those in whom subsequent weaning is
successful.66
Thus, unknown mechanisms associated with the act of spontaneous
breathing cause the worsening of respiratory mechanics in patients
who cannot be weaned from mechanical ventilation.
|
In addition to the increase in respiratory effort, an unsuccessful attempt
at spontaneous breathing causes considerable cardiovascular stress.67
Patients can have substantial increases in right and left
ventricular afterload, with increases of 39 and 27 percent in
pulmonary and systemic arterial pressures, respectively,64
most likely because the negative swings in intrathoracic pressure are
more extreme. At the completion of a trial of weaning, the level of
oxygen consumption is equivalent in patients who can be weaned and
in those who cannot. But how the cardiovascular system meets the
oxygen demand differs in the two groups of patients.64
In those who are successfully weaned, the oxygen demand is met
through an increase in oxygen delivery, mediated by the expected
increase in cardiac output on discontinuation of positive-pressure
ventilation. In patients who cannot be weaned, the oxygen demand is
met through an increase in oxygen extraction, and these patients
have a relative decrease in oxygen delivery.64
The greater oxygen extraction causes a substantial decrease in mixed
venous oxygen saturation, contributing to the arterial hypoxemia
that occurs in some patients.64
Over the course of a trial of spontaneous breathing, about half
of patients in whom the trial fails have an increase in carbon dioxide
tension of 10 mm Hg or more.7 The
hypercapnia is not usually a consequence of a decrease in minute
ventilation.63
Instead, hypercapnia results from rapid, shallow breathing, which
causes an increase in dead-space ventilation. In a small proportion
of patients who cannot be weaned, primary depression of respiratory
drive may be responsible for the hypercapnia.7
The discontinuation of mechanical ventilation needs to be
carefully timed. Premature discontinuation places severe stress on
the respiratory and cardiovascular systems, which can impede the
patient's recovery. Unnecessary delays in discontinuation can lead
to a host of complications. Decisions about timing that are based
solely on expert clinical judgment are frequently erroneous.68,69,70
Several functional measures are used to aid decision making. The
level of oxygenation must be satisfactory before one attempts to
discontinue mechanical ventilation. Yet in many patients with
satisfactory oxygenation, such attempts fail. The use of traditional
predictors of the success or failure of attempts — maximal
inspiratory pressure, vital capacity, and minute ventilation —
frequently has false positive or false negative results.71 A
more reliable predictor is the ratio of respiratory frequency to
tidal volume (f/VT).72
The ratio must be calculated during spontaneous breathing;
calculating it during pressure support markedly impairs its
predictive accuracy.68
The higher the ratio, the more severe the rapid, shallow breathing and
the greater the likelihood of unsuccessful weaning. A ratio of 100
best discriminates between successful and unsuccessful attempts at
weaning. In a case of clinical equipoise — that is, a pretest
probability of 50 percent — an f/VT of 80, which has a
likelihood ratio of 7.5, is associated with almost a 95 percent
post-test probability of successful weaning.73
If the f/VT is higher than 100, the likelihood ratio is 0.04
and the post-test probability of successful weaning is less than
5 percent.
Several groups of investigators have evaluated the predictive
value of f/VT.74,75,76,77,78
Its positive predictive value — the proportion of patients who are
successfully weaned among those for whom the ratio predicts success
— has generally been high (0.8 or higher). The negative predictive
value — the proportion of patients who cannot be weaned among those
for whom the ratio predicts failure — has sometimes been reported
to be low (0.5 or less). Low negative predictive values have often
been reported for patients with a high likelihood of successful
extubation — for example, patients undergoing routine postoperative
ventilatory assistance and patients who have tolerated initial
trials of weaning.75,76
There are four methods of weaning.79
The oldest method is to perform trials of spontaneous breathing several
times a day, with the use of a T-tube circuit containing an enriched
supply of oxygen. Initially 5 to 10 minutes in duration, the trials
are extended and repeated several times a day until the patient can
sustain spontaneous ventilation for several hours. This approach has
become unpopular because it requires considerable time on the part
of intensive care staff.
The two most common approaches, intermittent mandatory ventilation
and pressure support, decrease ventilatory assistance gradually by
respectively lowering the number of ventilator-assisted breaths or
the level of pressure. When a minimal level of ventilatory assistance
can be tolerated, the patient is extubated. The minimal level of
assistance, however, has never been well defined. For example,
pressure support of 6 to 8 cm of water is widely used to compensate
for the resistance imposed by the endotracheal tube and ventilator
circuit.80
A patient who can breathe comfortably at this level of pressure
support should be able to tolerate extubation. But if the upper
airways are swollen because an endotracheal tube has been in place
for several days, the work engendered by breathing through the
swollen airways is about the same as that caused by breathing
through an endotracheal tube.81
Accordingly, any amount of pressure support overcompensates and may
give misleading information about the likelihood that a patient can
tolerate extubation.
The fourth method of weaning is to perform a single daily T-tube
trial, lasting for up to two hours. If this trial is successful, the
patient is extubated; if the trial is unsuccessful, the patient is
given at least 24 hours of respiratory-muscle rest with full
ventilatory support before another trial is performed.82
Until the early 1990s, it was widely believed that all weaning
methods were equally effective, and the physician's judgment was
regarded as the critical determinant. But the results of randomized,
controlled trials clearly indicate that the period of weaning is as
much as three times as long with intermittent mandatory ventilation
as with trials of spontaneous breathing.61,62
In a study involving patients with respiratory difficulties on
weaning, trials of spontaneous breathing halved the weaning time as
compared with pressure support62;
in another study, the weaning time was similar with the two methods.61
Performing trials of spontaneous breathing once a day is as
effective as performing such trials several times a day62
but much simpler. In a recent study, half-hour trials of spontaneous
breathing were as effective as two-hour trials.83
However, this study involved all patients being considered for
weaning, not just those for whom there were difficulties with
weaning.
A two-stage approach to weaning — systematic measurement of
predictors, including f/VT , followed by a single daily trial of
spontaneous breathing — was compared with conventional management in
a randomized trial.69
Although the patients assigned to the two-stage approach were sicker
than those assigned to conventional weaning, they were weaned twice
as rapidly. The rate of complications and the costs of intensive
care were also lower with two-stage management than with
conventional management.
When patients can sustain spontaneous ventilation without undue
discomfort, they are extubated. About 10 to 20 percent of such patients
require reintubation.61,62
Mortality among patients who require reintubation is more than six
times as high as mortality among patients who can tolerate
extubation.83,84
The reason for the higher mortality is unknown; it is not clearly
related to the development of new problems after extubation or to
complications of reinserting the tube. Indeed, the need for
reintubation may simply be a marker of a more severe underlying
illness.
In a controlled trial involving patients who could not sustain
spontaneous ventilation, the patients who were extubated and then
received noninvasive ventilation through a face mask had a shorter
mean overall period of ventilatory support (10.2 days) than those
who remained intubated and were weaned by decreasing pressure
support (16.6 days).85
Although this result is promising, it is not clear how many such
patients or which ones could benefit from this approach.
Other Approaches to Mechanical Ventilation
Noninvasive ventilation, an approach that is becoming more
widespread, was reviewed in the Journal
in 1997.86
Two new approaches under investigation are liquid ventilation87
and proportional-assist ventilation16;
they have not yet been approved for general clinical use.
Conclusions
Since my previous overview of mechanical ventilation in the Journal, we have gained a better
understanding of the pathophysiology associated with unsuccessful
weaning and have learned how to wean patients more efficiently. We
have also learned how ventilator settings influence survival in
patients with the acute respiratory distress syndrome. Less progress
has been made in determining how the ventilator can best be used to
achieve maximal respiratory-muscle rest, which is the most common
reason for providing mechanical ventilation. Although further
research may lead to unexpected new insights, an important challenge
for researchers is to identify elements of our current knowledge
that can be incorporated into a clinical management scheme to
improve the outcome for patients who require ventilatory assistance.
Supported by a Merit Review grant from the Department of Veterans
Affairs Research and Development Service.
I am indebted to Drs. Amal Jubran, Franco Laghi, and Thomas Brack
for helpful criticisms on successive drafts of the manuscript.
Source Information
From the Division of Pulmonary and Critical Care Medicine, Edward
Hines, Jr., Veterans Affairs Hospital and Loyola University of Chicago Stritch
School of Medicine, Hines, Ill.
Address reprint requests to Dr. Tobin at the Division of Pulmonary
and Critical Care Medicine, Edward Hines, Jr., Veterans Affairs Hospital, Rte.
111N, Hines, IL 60141, or at [log in to unmask].
References
- Tobin
MJ. Mechanical ventilation. N Engl J Med 1994;330:1056-1061.
[Full Text]
- Esteban
A, Anzueto A, Alia I, et al. How is mechanical ventilation employed in the
intensive care unit? An international utilization review. Am J Respir Crit
Care Med 2000;161:1450-1458.
[Abstract/Full Text]
- Sassoon
CSH. Intermittent mandatory ventilation. In: Tobin JM, ed. Principles and
practice of mechanical ventilation. New York: McGraw-Hill, 1994:221-37.
- Marini
JJ, Smith TC, Lamb VJ. External work output and force generation during
synchronized intermittent mechanical ventilation: effect of machine
assistance on breathing effort. Am Rev Respir Dis 1988;138:1169-1179.
[Medline]
- Brochard
L. Pressure support ventilation. In: Tobin MJ, ed. Principles and practice
of mechanical ventilation. New York: McGraw-Hill, 1994:239-57.
- Jubran
A, Van de Graaff WB, Tobin MJ. Variability of patient-ventilator
interaction with pressure support ventilation in patients with chronic
obstructive pulmonary disease. Am J Respir Crit Care Med 1995;152:129-136.
[Abstract]
- Jubran
A, Tobin JM. Pathophysiologic basis of acute respiratory distress in
patients who fail a trial of weaning from mechanical ventilation. Am J
Respir Crit Care Med 1997;155:906-915.
[Abstract]
- Reid WD,
Huang J, Bryson S, Walker DC, Belcastro AN. Diaphragm injury and
myofibrillar structure induced by resistive loading. J Appl Physiol
1994;76:176-184.
[Medline]
- Leung P,
Jubran A, Tobin MJ. Comparison of assisted ventilator modes on triggering,
patient effort, and dyspnea. Am J Respir Crit Care Med 1997;155:1940-1948.
[Abstract]
- Anzueto
A, Peters JI, Tobin JM, et al. Effects of prolonged controlled mechanical
ventilation on diaphragmatic function in healthy adult baboons. Crit Care
Med 1997;25:1187-1190.
[Medline]
- Parthasarathy
S, Jubran A, Tobin MJ. Cycling of inspiratory and expiratory muscle groups
with the ventilator in airflow limitation. Am J Respir Crit Care Med
1998;158:1471-1478. [Erratum, Am J Respir Crit Care Med 1999;159:1023.]
[Abstract/Full Text]
- Simon
PM, Zurob AS, Wies WM, Leiter JC, Hubmayr RD. Entrainment of respiration
in humans by periodic lung inflations: effect of state and CO2.
Am J Respir Crit Care Med 1999;160:950-960.
[Abstract/Full Text]
- Parthasarathy
S, Jubran A, Tobin MJ. Assessment of neural inspiratory time in
ventilator-supported patients. Am J Respir Crit Care Med 2000;162:546-552.
[Abstract/Full Text]
- Sassoon
CSH, Gruer SE. Characteristics of the ventilator pressure- and
flow-trigger variables. Intensive Care Med 1995;21:159-168.
[Medline]
- Marini
JJ, Capps JS, Culver BH. The inspiratory work of breathing during assisted
mechanical ventilation. Chest 1985;87:612-618.
[Abstract]
- Giannouli
E, Webster K, Roberts D, Younes M. Response of ventilator-dependent
patients to different levels of pressure support and proportional assist.
Am J Respir Crit Care Med 1999;159:1716-1725.
[Abstract/Full Text]
- Sinderby
C, Navalesi P, Beck J, et al. Neural control of mechanical ventilation in
respiratory failure. Nat Med 1999;5:1433-1436.
[Medline]
- Puddy A,
Younes M. Effect of inspiratory flow rate on respiratory output in normal
subjects. Am Rev Respir Dis 1992;146:787-789.
[Medline]
- Laghi F,
Karamchandani K, Tobin MJ. Influence of ventilator settings in determining
respiratory frequency during mechanical ventilation. Am J Respir Crit Care
Med 1999;160:1766-1770.
[Abstract/Full Text]
- Pierson
DJ. Barotrauma and bronchopleural fistula. In: Tobin MJ, ed. Principles
and practice of mechanical ventilation. New York: McGraw-Hill,
1994:813-36.
- Webb HH,
Tierney DF. Experimental pulmonary edema due to intermittent positive
pressure ventilation with high inflation pressures: protection by positive
end-expiratory pressure. Am Rev Respir Dis 1974;110:556-565.
[Medline]
- Dreyfuss
D, Saumon G. Ventilator-induced lung injury: lessons from experimental
studies. Am J Respir Crit Care Med 1998;157:294-323.
[Full Text]
- Puybasset
L, Cluzel P, Chao N, Slutsky AS, Coriat P, Rouby JJ. A computed tomography
scan assessment of regional lung volume in acute lung injury. Am J Respir
Crit Care Med 1998;158:1644-1655.
[Abstract/Full Text]
- Gattinoni
L, Pesenti A, Bombino M, et al. Relationships between lung computed
tomographic density, gas exchange, and PEEP in acute respiratory failure.
Anesthesiology 1988;69:824-832.
[Medline]
- Rouby
JJ, Lherm T, Martin de Lassale E, et al. Histologic aspects of pulmonary
barotrauma in critically ill patients with acute respiratory failure.
Intensive Care Med 1993;19:383-389.
[Medline]
- Hickling
KG, Henderson SJ, Jackson R. Low mortality associated with low volume
pressure limited ventilation with permissive hypercapnia in severe adult
respiratory distress syndrome. Intensive Care Med 1990;16:372-377.
[Medline]
- Amato
MB, Barbas CS, Medeiros DM, et al. Beneficial effects of the "open
lung approach" with low distending pressures in acute respiratory
distress syndrome: a prospective randomized study on mechanical
ventilation. Am J Respir Crit Care Med 1995;152:1835-1846.
[Abstract]
- Amato
MBP, Barbas CSV, Medeiros DM, et al. Effect of a protective-ventilation
strategy on mortality in the acute respiratory distress syndrome. N Engl J
Med 1998;338:347-354.
[Abstract/Full Text]
- The
Acute Respiratory Distress Syndrome Network. Ventilation with lower tidal
volumes as compared with traditional tidal volumes for acute lung injury
and the acute respiratory distress syndrome. N Engl J Med
2000;342:1301-1308.
[Abstract/Full Text]
- Stewart
TE, Meade MO, Cook DJ, et al. Evaluation of a ventilation strategy to
prevent barotrauma in patients at high risk for acute respiratory distress
syndrome. N Engl J Med 1998;338:355-361.
[Abstract/Full Text]
- Brochard
L, Roudot-Thoraval F, Roupie E, et al. Tidal volume reduction for
prevention of ventilator-induced lung injury in acute respiratory distress
syndrome. Am J Respir Crit Care Med 1998;158:1831-1838.
[Abstract/Full Text]
- Brower
RG, Shanholtz CB, Fessler HE, et al. Prospective, randomized, controlled
clinical trial comparing traditional versus reduced tidal volume
ventilation in acute respiratory distress syndrome patients. Crit Care Med
1999;27:1492-1498.
[Medline]
- Tobin
MJ. Culmination of an era in research on the acute respiratory distress
syndrome. N Engl J Med 2000;342:1360-1361.
[Full Text]
- Brochard
L. Respiratory pressure-volume curves. In: Tobin MJ, ed. Principles and
practice of intensive care monitoring. New York: McGraw-Hill,
1998:597-616.
- Roupie
E, Dambrosio M, Servillo G, et al. Titration of tidal volume and induced
hypercapnia in acute respiratory distress syndrome. Am J Respir Crit Care
Med 1995;152:121-128.
[Abstract]
- Richecoeur
J, Lu Q, Vieira SR, et al. Expiratory washout versus optimization of
mechanical ventilation during permissive hypercapnia in patients with
severe acute respiratory distress syndrome. Am J Respir Crit Care Med
1999;160:77-85.
[Abstract/Full Text]
- Feihl F,
Perret C. Permissive hypercapnia: how permissive should we be? Am J Respir
Crit Care Med 1994;150:1722-1737.
[Medline]
- Feihl F,
Eckert P, Brimioulle S, et al. Permissive hypercapnia impairs pulmonary
gas exchange in the acute respiratory distress syndrome. Am J Respir Crit
Care Med 2000;162:209-215.
[Abstract/Full Text]
- Mead J,
Collier C. Relation of volume history of lungs to respiratory mechanics in
anesthetized dogs. J Appl Physiol 1959;14:669-678.
- Pelosi
P, Cadringher P, Bottino N, et al. Sigh in acute respiratory distress
syndrome. Am J Respir Crit Care Med 1999;159:872-880.
[Abstract/Full Text]
- Dambrosio
M, Roupie E, Mollett JJ, et al. Effects of positive end-expiratory
pressure and different tidal volumes on alveolar recruitment and
hyperinflation. Anesthesiology 1997;87:495-503.
[Medline]
- Vieira
SR, Puybasset L, Lu Q, et al. A scanographic assessment of pulmonary
morphology in acute lung injury: significance of the lower inflection
point detected on the lung pressure-volume curve. Am J Respir Crit Care
Med 1999;159:1612-1623.
[Abstract/Full Text]
- Vieira
SR, Puybasset L, Richecoeur J, et al. A lung computed tomographic
assessment of positive end-expiratory pressure-induced lung
overdistension. Am J Respir Crit Care Med 1998;158:1571-1577.
[Abstract/Full Text]
- Horton
WG, Cheney FW. Variability of effect of positive end expiratory pressure.
Arch Surg 1975;110:395-398.
[Medline]
- Kanarek
DJ, Shannon DC. Adverse effect of positive end-expiratory pressure on
pulmonary perfusion and arterial oxygenation. Am Rev Respir Dis
1975;112:457-459.
[Medline]
- Gattinoni
L, Pelosi P, Suter PM, Pedoto A, Vercesi P, Lissoni A. Acute respiratory
distress syndrome caused by pulmonary and extrapulmonary disease:
different syndromes? Am J Respir Crit Care Med 1998;158:3-11.
[Abstract/Full Text]
- Lu Q,
Vieira SR, Richecoeur J, et al. A simple automated method for measuring
pressure-volume curves during mechanical ventilation. Am J Respir Crit
Care Med 1999;159:275-282.
[Abstract/Full Text]
- Argiras
EP, Blakeley CR, Dunnill MS, Otremski S, Sykes MK. High PEEP decreases
hyaline membrane formation in surfactant deficient lungs. Br J Anaesth
1987;59:1278-1285.
[Abstract]
- Tremblay
L, Valenza F, Ribeiro SP, Li J, Slutsky AS. Injurious ventilatory
strategies increase cytokines and c-fos m-RNA expression in an isolated
rat lung model. J Clin Invest 1997;99:944-952.
[Abstract/Full Text]
- Mead J,
Takishima T, Leith D. Stress distribution in lungs: a model of pulmonary
elasticity. J Appl Physiol 1970;28:596-608.
[Medline]
- Hickling
KG. The pressure-volume curve is greatly modified by recruitment: a mathematical
model of ARDS lungs. Am J Respir Crit Care Med 1998;158:194-202.
[Abstract/Full Text]
- Martynowicz
MA, Minor TA, Walters BJ, Hubmayr RD. Regional expansion of oleic
acid-injured lungs. Am J Respir Crit Care Med 1999;160:250-258.
[Abstract/Full Text]
- Lichtwarck-Aschoff
M, Mols G, Hedlund AJ, et al. Compliance is nonlinear over tidal volume
irrespective of positive end-expiratory pressure level in
surfactant-depleted piglets. Am J Respir Crit Care Med 2000;162:2125-2133.
[Abstract/Full Text]
- Carney
DE, Bredenberg CE, Schiller HJ, et al. The mechanism of lung volume change
during mechanical ventilation. Am J Respir Crit Care Med
1999;160:1697-1702.
- Harris
RS, Hess DR, Venegas JG. An objective analysis of the pressure-volume
curve in the acute respiratory distress syndrome. Am J Respir Crit Care
Med 2000;161:432-439.
[Abstract/Full Text]
- Pelosi
P, Tubiolo D, Mascheroni D, et al. Effects of the prone position on
respiratory mechanics and gas exchange during acute lung injury. Am J
Respir Crit Care Med 1998;157:387-393.
[Abstract/Full Text]
- Gattinoni
L, Pelosi P, Vitale G, Pesenti A, D'Andrea L, Mascheroni D. Body position
changes redistribute lung computed-tomographic density in patients with
acute respiratory failure. Anesthesiology 1991;74:15-23.
[Medline]
- Mutoh T,
Guest RJ, Lamm WJ, Albert RK. Prone position alters the effect of volume
overload on regional pleural pressures and improves hypoxemia in pigs in vivo.
Am Rev Respir Dis 1992;146:300-306.
[Medline]
- Mure M,
Domino KB, Lindahl SG, Hlastala MP, Altemeier WA, Glenny RW. Regional
ventilation-perfusion distribution is more uniform in the prone position.
J Appl Physiol 2000;88:1076-1083.
[Abstract/Full Text]
- Albert
RK, Hubmayr RD. The prone position eliminates compression of the lungs by
the heart. Am J Respir Crit Care Med 2000;161:1660-1665.
[Abstract/Full Text]
- Brochard
L, Rauss A, Benito S, et al. Comparison of three methods of gradual
withdrawing from ventilatory support during weaning from mechanical
ventilation. Am J Respir Crit Care Med 1994;150:896-903.
[Abstract]
- Esteban
A, Frutos F, Tobin MJ, et al. A comparison of four methods of weaning
patients from mechanical ventilation. N Engl J Med 1995;332:345-350.
[Abstract/Full Text]
- Tobin
MJ, Perez W, Guenther SM, et al. The pattern of breathing during
successful and unsuccessful trials of weaning from mechanical ventilation.
Am Rev Respir Dis 1986;134:1111-1118.
[Medline]
- Tobin
MJ, Chadha TS, Jenouri G, Birch SJ, Gazeroglu HB, Sackner MA. Breaking
patterns. 1. Normal subjects. Chest 1983;84:202-205.
[Abstract]
- Jubran
A, Mathru M, Dries D, Tobin MJ. Continuous recordings of mixed venous
oxygen saturation during weaning from mechanical ventilation and the
ramifications thereof. Am J Respir Crit Care Med 1998;158:1763-1769.
[Abstract/Full Text]
- Jubran
A, Tobin MJ. Passive mechanics of lung and chest wall in patients who
failed or succeeded in trials of weaning. Am J Respir Crit Care Med 1997;155:916-921.
[Abstract]
- Lemaire
F, Teboul JL, Cinotti L, et al. Acute left ventricular dysfunction during
unsuccessful weaning from mechanical ventilation. Anesthesiology
1988;69:171-179.
[Medline]
- Stroetz
RW, Hubmayr RD. Tidal volume maintenance during weaning with pressure
support. Am J Respir Crit Care Med 1995;152:1034-1040.
[Abstract]
- Ely EW,
Baker AM, Dunagan DP, et al. Effect on the duration of mechanical
ventilation of identifying patients capable of breathing spontaneously. N
Engl J Med 1996;335:1864-1869.
[Abstract/Full Text]
- Coplin
WM, Pierson DJ, Cooley KD, Newell DW, Rubenfeld GD. Implications of
extubation delay in brain-injured patients meeting standard weaning
criteria. Am J Respir Crit Care Med 2000;161:1530-1536.
[Abstract/Full Text]
- Tobin
MJ, Alex CG. Discontinuation of mechanical ventilation. In: Tobin MJ, ed.
Principles and practice of mechanical ventilation. New York: McGraw-Hill,
1994:1177-206.
- Yang KL,
Tobin MJ. A prospective study of indexes predicting the outcome of trials
of weaning from mechanical ventilation. N Engl J Med 1991;324:1445-1450.
[Abstract]
- Jaeschke
RZ, Meade MO, Guyatt GH, Keenan SP, Cook DJ. How to use diagnostic test
articles in the intensive care unit: diagnosing weanability using f/Vt.
Crit Care Med 1997;25:1514-1521.
[Medline]
- Sassoon
CSH, Mahutte CK. Airway occlusion pressure and breathing pattern as
predictors of weaning outcome. Am Rev Respir Dis 1993;148:860-866.
[Medline]
- Epstein
SK. Etiology of extubation failure and the predictive value of the rapid
shallow breathing index. Am J Respir Crit Care Med 1995;152:545-549.
[Abstract]
- Jacob B,
Chatila W, Manthous CA. The unassisted respiratory rate/tidal volume ratio
accurately predicts weaning outcome in postoperative patients. Crit Care
Med 1997;25:253-257.
[Medline]
- Vallverdu
I, Calaf N, Subirana M, Net A, Benito S, Mancebo J. Clinical
characteristics, respiratory functional parameters, and outcome of a
two-hour T-piece trial in patients weaning from mechanical ventilation. Am
J Respir Crit Care Med 1998;158:1855-1862.
[Abstract/Full Text]
- Maldonado
A, Bauer TT, Ferrer M, et al. Capnometric recirculation gas tonometry and
weaning from mechanical ventilation. Am J Respir Crit Care Med
2000;161:171-176.
[Abstract/Full Text]
- Tobin
MJ. 1999 Donald F Egan Scientific Lecture: weaning from mechanical
ventilation: what have we learned? Respir Care 2000;45:417-431.
[Medline]
- Brochard
L, Rua F, Lorino H, Lemaire F, Harf A. Inspiratory pressure support
compensates for the additional work of breathing caused by the
endotracheal tube. Anesthesiology 1991;75:739-745.
[Medline]
- Straus
C, Louis B, Isabey D, Lemaire F, Harf A, Brochard L. Contribution of the
endotracheal tube and the upper airway to breathing workload. Am J Respir
Crit Care Med 1998;157:23-30.
[Abstract/Full Text]
- Laghi F,
D'Alfonso N, Tobin MJ. Pattern of recovery from diaphragmatic fatigue over
24 hours. J Appl Physiol 1995;79:539-546.
[Medline]
- Esteban
A, Alia I, Tobin MJ, et al. Effect of spontaneous breathing trial duration
on outcome of attempts to discontinue mechanical ventilation. Am J Respir
Crit Care Med 1999;159:512-518.
[Abstract/Full Text]
- Epstein
SK, Ciubotaru RL, Wong JB. Effect of failed extubation on the outcome of
mechanical ventilation. Chest 1997;112:186-192.
[Abstract]
- Nava S,
Ambrosino N, Clini E, et al. Noninvasive mechanical ventilation in the
weaning of patients with respiratory failure due to chronic obstructive
pulmonary disease: a randomized, controlled trial. Ann Intern Med
1998;128:721-728.
[Medline]
- Hillberg
RE, Johnson DC. Noninvasive ventilation. N Engl J Med 1997;337:1746-1752.
[Full Text]
- Leach
CL, Greenspan JS, Rubenstein SD, et al. Partial liquid ventilation with
perflubron in premature infants with severe respiratory distress syndrome.
N Engl J Med 1996;335:761-767.
[Abstract/Full Text]
Edward E.
Rylander, M.D.
Diplomat American
Board of Family Practice.
Diplomat American
Board of Palliative Medicine.