|
|
The Interval between Pregnancies and the Risk of
Preeclampsia
Rolv Skjærven, Ph.D., Allen J. Wilcox, M.D., Ph.D., and Rolv
T. Lie, Ph.D.
ABSTRACT
Background The risk of
preeclampsia is generally lower in second pregnancies than in first
pregnancies, but not if the mother has a new partner for the second pregnancy.
One explanation is that the risk is reduced with repeated maternal
exposure and adaptation to specific antigens from the same partner.
However, the difference in risk might instead be explained by the
interval between births. A longer interbirth interval may be
associated with both a change of partner and a higher risk of
preeclampsia.
Methods We used data
from the Medical Birth Registry of Norway, a population-based
registry that includes births that occurred between 1967 and 1998. We
studied 551,478 women who had two or more singleton deliveries and
209,423 women who had three or more singleton deliveries.
Results Preeclampsia
occurred during 3.9 percent of first pregnancies, 1.7 percent of
second pregnancies, and 1.8 percent of third pregnancies when the
woman had the same partner. The risk in a second or third pregnancy
was directly related to the time that had elapsed since the
preceding delivery, and when the interbirth interval was 10 years or
more, the risk approximated that among nulliparous women. After
adjustment for the presence or absence of a change of partner,
maternal age, and year of delivery, the odds ratio for preeclampsia
for each one-year increase in the interbirth interval was 1.12 (95
percent confidence interval, 1.11 to 1.13). In unadjusted analyses,
a pregnancy involving a new partner was associated with higher risk
of preeclampsia, but after adjustment for the interbirth interval,
the risk of preeclampsia was reduced (odds ratio for preeclampsia
with a change of partner, 0.73; 95 percent confidence interval, 0.66
to 0.81).
Conclusions The
protective effect of previous pregnancy against preeclampsia is
transient. After adjustment for the interval between births, a
change of partner is not associated with an increased risk of
preeclampsia.
Preeclampsia is a transient but
potentially dangerous complication of pregnancy that affects 3 to 5
percent of pregnant women.1,2
Although the causes of preeclampsia remain uncertain,3
epidemiologic features of the condition have led to speculation
about immunologic causes. The risk of preeclampsia is at least twice
as high during first pregnancies as during second or later
pregnancies.4,5,6
Recent studies have suggested that the risk may decrease with a
second pregnancy only if the mother's partner is the same.1,7
The hypothesis is that the risk of preeclampsia may be reduced with
repeated maternal exposure and adaptation to specific foreign antigens
of the partner.3,8,9
According to this hypothesis, a new partner presents new antigens,
which results in a risk of preeclampsia that is similar to the risk
during a first pregnancy. However, it is also possible that the
increased risk of preeclampsia associated with a change of partner
might be attributable to a longer interval since the previous
delivery, which may also increase the risk of preeclampsia.5,10 We
used a large registry in Norway to evaluate the effects on the risk
of preeclampsia of both the interbirth interval and a change of
partner.
Methods
Data on Deliveries
We used data from the Medical Birth Registry of Norway, comprising
the records of more than 1.8 million births between 1967 and 1998.
Stillbirths occurring after 16 weeks or more of gestation are also
reported to the registry. The registry contains a unique personal
identification number for all mothers, all liveborn children, and
nearly all fathers.
All children born to a given woman were linked by means of the
national identification number of the woman. We identified 551,478 sets
(7.6 percent) of first and second singleton deliveries that occurred
during this period. Of these, 509,548 of the pairs (92.4 percent)
had the same father, and 31,683 pairs (5.7 percent) had different
fathers; for the remaining 10,247 pairs (1.9 percent), it could not
be determined whether the father was the same. Similarly, we
identified 209,423 sets of first, second, and third singleton
deliveries. Of these, 158,284 sets (7.6 percent) had the same
father; in the case of 24,252 sets (11.6 percent), the partner had
changed either between the first and second deliveries (4.6 percent)
or between the second and third deliveries (7.0 percent). For the
remaining 26,887 sets (12.8 percent), information on the father was
missing for at least one pregnancy.
Calculation of Interbirth Interval
The interbirth interval was calculated as the time (in days) between
two consecutive birth dates. We used birth dates (rather than
approximate dates of conception) for calculating the interbirth interval
because this information was virtually 100 percent complete, whereas
information on the gestational age (which is necessary for
estimating the date of conception) was more often missing or
unreliable. The effect of using the birth date rather than the
estimated date of conception was expected to be minimal for the
present analysis, which focuses on longer interbirth intervals. The
interbirth interval was categorized according to completed years
(e.g., an interbirth interval of two years indicates a period of at
least two years and less than three years).
Definition of Preeclampsia
Preeclampsia was defined as an increase in blood pressure to at
least 140/90 mm Hg after the 20th week of gestation, an increase in
diastolic blood pressure of at least 15 mm Hg from the level measured
before the 20th week, or an increase in systolic blood pressure of
at least 30 mm Hg from the level measured before the 20th week,
combined with proteinuria (protein excretion, at least 0.3 g per 24
hours).11
A diagnosis of preeclampsia in the medical record is routinely
entered on the medical registration form as a specified diagnosis by
the midwife or obstetrician. In some cases, the registration form
contains information on the presence of hypertension, proteinuria,
or edema during pregnancy. We included as cases of preeclampsia all
pregnancies with a specified diagnosis of preeclampsia and
pregnancies with a combination of pregnancy-related hypertension and
proteinuria.5
Statistical Analysis
We used stratification and logistic-regression techniques to evaluate
possible confounding. In these analyses, we divided the deliveries
into three periods according to the year (1967 to 1976, 1977 to
1986, and 1987 to 1998) and maternal age into five categories (less
than 20 years, 20 to 24 years, 25 to 29 years, 30 to 34 years, and
35 years or more). In assessing the interbirth interval, we used the
mother's age at the time of the later delivery and the period during
which that delivery occurred. Age and period were treated as
categorical variables, and the interval between pregnancies was
treated as a linear variable, categorized into 10 groups according
to completed years (with <1 year included in the 1-year category
and >10 years included in the 10-year category). For the
interbirth interval, the main variable of interest, we report the
increase in risk with each additional year between deliveries as an
estimated odds ratio. For most analyses, the data were restricted to
women with no history of preeclampsia during previous pregnancies
and to the pregnancies of women who had the same partner for all
pregnancies. We only considered pregnancies that led to a recorded birth,
including stillbirths that occurred after at least 16 weeks of
gestation.
Results
Risk of Preeclampsia According to Parity
Preeclampsia occurred during 3.9 percent of first pregnancies.
When subsequent pregnancies involved the same partner, preeclampsia occurred
during 1.7 percent of second pregnancies and 1.8 percent of third
pregnancies (Table
1). When women with previous preeclampsia were excluded,
preeclampsia occurred during only 1.3 percent of second and third
pregnancies (Table
1).
|
The Effect of the Interval between Deliveries
Among women with no history of preeclampsia, the median interbirth
interval was 2.9 years between the first and the second deliveries and
3.6 years between the second and the third deliveries. The risk of preeclampsia
during the second pregnancy was found to increase steadily as the
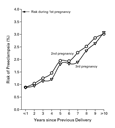
time since the first delivery increased (Figure 1). The
estimated odds ratio for preeclampsia was 1.16 per additional year
(95 percent confidence interval, 1.15 to 1.18). By 10 years after
the first pregnancy, the risk of preeclampsia had more than tripled,
nearly reaching the level of the risk found among nulliparous women.
An increasing interval between the second and the third deliveries
was similarly associated with an increasing risk of preeclampsia (Figure 1).
|
The risk of preeclampsia is known to increase with maternal age,12
and this relation might contribute to an apparent increase in risk with
an increasing interbirth interval. Nonetheless, the increased risk
of preeclampsia associated with an increasing interbirth interval
remained after we controlled for maternal age (in five-year
categories) (odds ratio, 1.13 per year; 95 percent confidence
interval, 1.12 to 1.14). An analysis that adjusted for maternal age
with the use of one-year age categories gave the same results.
There was a moderate increase in the risk of preeclampsia during
the first 15 years of the birth registry (1967 to 1982), and the
risk remained stable thereafter. When we adjusted for the year of
delivery, the results were essentially unchanged (data not shown).
Finally, we considered the possibility that the increase in the
risk of preeclampsia with an increasing interbirth interval could be
confounded by an association between preeclampsia and subfertility.
If less fertile women are at higher risk for preeclampsia, then the
increase in the risk of preeclampsia with an increasing interbirth
interval could be attributable to the overrepresentation of less
fertile women. However, we found no association between the risk of
preeclampsia in the first pregnancy and the interval between the
first and second pregnancies (odds ratio for preeclampsia in the
first pregnancy, 1.01 per year; 95 percent confidence interval, 0.99
to 1.01).
Effect of a Change of Partner
The association between an increasing interbirth interval and
an increasing risk of preeclampsia might be explained by the fact
that a change of partner is more common among the women with longer
interbirth intervals. In our cohort, about 6 percent of the women
changed their partners between their first and second pregnancies.
The median time to the second delivery when the partner remained the
same was 2.9 years, as compared with 5.9 years with a change of
partner (Table 2).
Similarly, the median time from the second to the third delivery was
3.6 years when the partner remained the same and 7.5 years with a
change of partner. Among women with no history of preeclampsia, the
complication occurred during 1.3 percent of second pregnancies for
which the partner remained the same and 1.5 percent of those for
which there was a change of partner. This excess risk of preeclampsia
for women who changed partners as compared with those who remained
with the same partner was small but statistically significant (odds
ratio, 1.14; 95 percent confidence interval, 1.04 to 1.26) (Table 3). The
risk of preeclampsia was similarly increased for the third pregnancy
if there was a different partner (odds ratio, 1.42; 95 percent
confidence interval, 1.25 to 1.62).
|
|
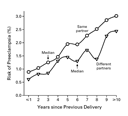
We assessed the association between the interbirth interval and the risk
of preeclampsia during the second pregnancy, stratifying the women
according to whether or not their partner was the same for both
pregnancies. A strong effect of the interbirth interval was seen
regardless of change or lack of change of partner (Figure 2). The
effect of changing the partner was eliminated when the interbirth
interval was taken into account. In fact, after the interval had
been taken into account, the risk of preeclampsia was lower for
pregnancies involving a new partner than for those involving the
same partner (Figure
2).
|
We also used multivariate analysis to assess simultaneously the
effects of the interbirth interval and change or lack of change of
partner on the risk of preeclampsia (Table 3). After
adjustment for the interbirth interval, a change of partner was
no longer associated with an increased risk of preeclampsia but, rather,
with a significantly decreased risk of this complication. The
results were essentially the same after further adjustment for
maternal age and the period when the delivery occurred (Table 3).
These analyses included stillbirths that had occurred as early
as 16 weeks after gestation began. Since preeclampsia typically occurs
later in pregnancy, we reanalyzed the data for second deliveries excluding
stillbirths that had occurred at less than 21 weeks of gestation.
The associations of the interbirth interval and a change of partner
with the risk of preeclampsia were materially unchanged.
Discussion
We found that multiparous women who are pregnant 10 years or more
after their previous pregnancy are as likely to have preeclampsia as
nulliparous women. Preeclampsia has been described as "a disease
of first pregnancy"3,13,14
and is sometimes defined as occurring only among nulliparous women.
Although our data confirm that the risk of preeclampsia falls
sharply after the first pregnancy, we also found that the risk
subsequently increases over time. This striking increase in risk
with an increasing interbirth interval suggests that the benefit of
higher parity in terms of the risk of preeclampsia is only
transient.
A change of partner has been thought to increase the risk of preeclampsia.1,7,13,15
This observation is likely to be confounded by the effect of the
interbirth interval. The apparent increase in the risk of
preeclampsia with a change of partner has been interpreted as
support for the hypothesis that a failure of the immune system to
adapt to the partner's antigens causes preeclampsia. This theory was
proposed in 1975 in a case report of preeclampsia during the second
pregnancy of a woman who changed her partner after having her first
child.16
This has led to extensive speculation about possible immune
mechanisms related to a change of partner.3,8,9,14,16,17,18
Our data suggest that after appropriate adjustment for the
interbirth interval, there is no increased risk associated with a
change of partner. The observed association between the interbirth
interval and the risk of preeclampsia may be relevant in
interpreting the results of other studies of risk factors for
preeclampsia. For example, miscarriage has been associated with a
reduction in the risk of preeclampsia in a subsequent pregnancy.4,12,19
This result might be explained by the fact that the average interval
between pregnancies is shorter after a miscarriage than after a live
birth.20
Also, artificial insemination with donor semen and the use of donated
oocytes have been reported to increase the risk of preeclampsia.21,22
Women who receive these treatments have often been trying for a long
time to become pregnant.
Although the influence of the time since a previous pregnancy
appears to be as strong as that of any of the known risk factors for
preeclampsia, the recognition of this association should not be taken
as a recommendation of short intervals between pregnancies. Other
adverse outcomes of pregnancy — most notably preterm delivery — are
more likely with very short intervals.23 In
terms of these other adverse outcomes, the lower limit of the
interval at which high risk is attenuated varies, ranging from 6
months between delivery and subsequent conception in developed
countries to 18 months in developing countries.24
Our study has some limitations. We do not have data on the smoking
status of the women, which may confound the association between a
change of partner and the risk of preeclampsia. Many pregnant women
in Norway smoke,25,26
and smoking may be more common among women whose marriages end in
divorce.27
If so, smoking may be more frequent among women who later become
pregnant by a new partner. Since smoking is associated with a
reduction in the risk of preeclampsia,4,12,28
this set of associations might explain the lower risk of
preeclampsia observed among women who change partners.
Another limitation is the lack of data on obesity. Obesity is
associated with an increased risk of preeclampsia.4,12
The risk of obesity increases with a woman's age and parity.
However, if increasing weight were contributing to the effect of the
interbirth interval, we would have expected a higher risk of preeclampsia
during the third pregnancy than during the second pregnancy. No such
increase was seen (Figure
1).
An extended interval between pregnancies appears to be a major
risk factor for preeclampsia, with the risk after 10 years similar to
that among nulliparous women. Furthermore, after adjustment for the
interbirth interval, a change of partner between one pregnancy and
the next is not a risk factor for preeclampsia.
Supported by
funds from the Norwegian Foundation for Health and Rehabilitation.
We are indebted to Dr. Donna Baird, Dr. Grace Egeland, Dr. Lorentz
M. Irgens, Dr. Kari Klungsøyr Melve, Dr. Svein Rasmussen, and Dr.
Clarice Weinberg for their constructive comments on previous
versions of the manuscript.
Source Information
From the Section for Medical Statistics, Department of Public
Health and Primary Health Care, and the Medical Birth Registry of Norway, Locus
for Registry-Based Epidemiology, University of Bergen, Bergen, Norway (R.S.,
R.T.L.); and the Epidemiology Branch, National Institute of Environmental
Health Sciences, Research Triangle Park, N.C. (A.J.W.).
Address reprint requests to Professor Skjærven at the Section for
Medical Statistics, University of Bergen, Armauer Hansens Bldg., 5021 Bergen,
Norway, or at [log in to unmask].
References
- Trupin
LS, Simon LP, Eskenazi B. Change in paternity: a risk factor for
preeclampsia in multiparas. Epidemiology 1996;7:240-244.
[Medline]
- Roberts
JM, Cooper DW. Pathogenesis and genetics of pre-eclampsia. Lancet 2001;357:53-56.
[Medline]
- Dekker
GA, Sibai BM. Etiology and pathogenesis of preeclampsia: current concepts.
Am J Obstet Gynecol 1998;179:1359-1375.
[Medline]
- Eskenazi
B, Fenster L, Sidney S. A multivariate analysis of risk factors for
preeclampsia. JAMA 1991;266:237-241.
[Medline]
- Lie RT,
Rasmussen S, Brunborg H, Gjessing HK, Lie-Nielsen E, Irgens LM. Fetal and
maternal contributions to risk of pre-eclampsia: population based study.
BMJ 1998;316:1343-1347.
[Abstract/Full Text]
- Pipkin
FB. Risk factors for preeclampsia. N Engl J Med 2001;344:925-926.
[Full Text]
- Robillard
PY, Hulsey TC, Alexander GR, Keenan A, de Caunes F, Papiernik E. Paternity
patterns and risk of preeclampsia in the last pregnancy in multiparae. J
Reprod Immunol 1993;24:1-12.
[Medline]
- Dekker
GA, Robillard PY, Hulsey TC. Immune maladaptation in the etiology of
preeclampsia: a review of corroborative epidemiologic studies. Obstet
Gynecol Surv 1998;53:377-382.
[Medline]
- Dekker
G, Sibai BM. Primary, secondary, and tertiary prevention of pre-eclampsia.
Lancet 2001;357:209-215.
[Medline]
- Conde-Agudelo
A, Belizan JM. Maternal morbidity and mortality associated with
interpregnancy interval: cross sectional study. BMJ 2000;321:1255-1259.
[Abstract/Full Text]
- Lindheimer
MD, Roberts JM, Cunningham FG, Chesley L. Introduction, history,
controversies, and definitions. In: Lindheimer MD, Roberts JM, Cunningham
FG, eds. Chesley's hypertensive disorders in pregnancy. 2nd ed. Stamford,
Conn.: Appleton & Lange, 1999:3-41.
- Sibai
BM, Ewell M, Levine RJ, et al. Risk factors associated with preeclampsia
in healthy nulliparous women. Am J Obstet Gynecol 1997;177:1003-1010.
[Medline]
- Robillard
PY, Dekker GA, Hulsey TC. Revisiting the epidemiological standard of
preeclampsia: primigravidity or primipaternity? Eur J Obstet Gynecol
Reprod Biol 1999;84:37-41.
[Medline]
- Tubbergen
P, Lachmeijer AM, Althuisius SM, Vlak ME, van Geijn HP, Dekker GA. Change
in paternity: a risk factor for preeclampsia in multiparous women? J
Reprod Immunol 1999;45:81-88.
[Medline]
- Feeney
JG, Scott JS. Pre-eclampsia and changed paternity. Eur J Obstet Gynecol
Reprod Biol 1980;11:35-38.
[Medline]
- Need JA.
Pre-eclampsia in pregnancies by different fathers: immunological studies.
Br Med J 1975;1:548-549.
[Medline]
- Need JA.
Some immunological aspects of preeclampsia. Perspect Nephrol Hypertens
1976;5:169-176.
[Medline]
- Dekker
GA, Sibai BM. The immunology of preeclampsia. Semin Perinatol
1999;23:24-33.
[Medline]
- Eras JL,
Saftlas AF, Triche E, Hsu CD, Risch HA, Bracken MB. Abortion and its effect
on risk of preeclampsia and transient hypertension. Epidemiology
2000;11:36-43.
[Medline]
- Basso O,
Olsen J, Christensen K. Risk of preterm delivery, low birthweight and
growth retardation following spontaneous abortion: a registry-based study
in Denmark. Int J Epidemiol 1998;27:642-646.
[Abstract]
- Need JA,
Bell B, Meffin E, Jones WR. Pre-eclampsia in pregnancy from donor
inseminations. J Reprod Immunol 1983;5:329-338.
[Medline]
- Smith
GN, Walker M, Tessier JL, Millar KG. Increased incidence of preeclampsia
in women conceiving by intrauterine insemination with donor versus partner
sperm for treatment of primary infertility. Am J Obstet Gynecol
1997;177:455-458.
[Medline]
- Zhu B-P,
Rolfs RT, Nangle BE, Horan JM. Effect of the interval between pregnancies
on perinatal outcomes. N Engl J Med 1999;340:589-594.
[Abstract/Full Text]
- Rawlings
JS, Rawlings VB, Read JA. Prevalence of low birth weight and preterm
delivery in relation to the interval between pregnancies among white and
black women. N Engl J Med 1995;332:69-74.
[Abstract/Full Text]
- Haug K,
Irgens LM, Baste V, Markestad T, Skjaerven R, Schreuder P. Secular trends
in breastfeeding and parental smoking. Acta Paediatr 1998;87:1023-1027.
[Medline]
- Haug K,
Irgens LM, Skjaerven R, Markestad T, Baste V, Schreuder P. Maternal
smoking and birthweight: effect modification of period, maternal age and
paternal smoking. Acta Obstet Gynecol Scand 2000;79:485-489.
[Medline]
- Fu H,
Goldman N. The association between health-related behaviours and the risk
of divorce in the USA. J Biosoc Sci 2000;32:63-88.
[Medline]
- Duffus
GM, MacGillivray I. The incidence of pre-eclamptic toxaemia in smokers and
non-smokers. Lancet 1968;1:994-995.
[Medline]
Edward E.
Rylander, M.D.
Diplomat American
Board of Family Practice.
Diplomat American
Board of Palliative Medicine.