|
|
Migraine — Current Understanding and Treatment
Peter J. Goadsby, M.D., D.Sc., Richard B. Lipton, M.D., and
Michel D. Ferrari, M.D., Ph.D.
|
|
Migraine is a common, chronic, incapacitating neurovascular disorder,
characterized by attacks of severe headache, autonomic nervous
system dysfunction, and in some patients, an aura involving neurologic
symptoms.1,2
Recent advances in basic and applied clinical neuroscience3
have led to the development of a new class of selective serotonin
(5-hydroxytryptamine [5-HT]) receptor agonists that activate 5-HT1B
and 5-HT1D (5-HT1B/1D) receptors and are
known as the triptans; these agents have changed the lives of
countless patients with migraine. Despite such progress, migraine
remains underdiagnosed and the available therapies underused.4 In
this article, we review the current understanding of the
epidemiology, pathophysiology, and treatment of migraine.
Clinical Manifestations
Migraine is characterized by episodes of head pain that is often
throbbing and frequently unilateral and may be severe. In migraine without
aura (previously known as common migraine), attacks are usually
associated with nausea, vomiting, or sensitivity to light, sound, or
movement.5
When untreated, these attacks typically last 4 to 72 hours.6 A
combination of features is required for the diagnosis, but not all
features are present in every attack or in every patient (Table 1).
|
These symptoms distinguish migraine from tension-type headache, the
most common form of primary headache, which is characterized by the
lack of associated features. Any severe and recurrent headache is
most likely to be a form of migraine and to be responsive to
antimigraine therapy.8 In
15 percent of patients, migraine attacks are usually preceded or
accompanied by transient focal neurologic symptoms, which are
usually visual; such patients have migraine with aura (previously
known as classic migraine).9
In a recent large, population-based study, 64 percent of patients with
migraine had only migraine without aura, 18 percent had only
migraine with aura, and 13 percent had both types of migraine (the
remaining 5 percent had aura without headache). Thus, up to 31
percent of patients with migraine have aura on some occasions,10
but clinicians who rely on the presence of aura for the diagnosis of
migraine will miss many cases.
We find it useful to assess the severity and effects of migraine
by asking about time lost because of migraine at work or school, in
performing household work or chores, or in family, social, and
leisure activities. One can ask patients directly about temporary
disability, have them keep a diary, or get a quick but accurate
estimate with the use of the Migraine Disability Assessment Scale
(MIDAS) (Table 2),
a well-validated five-item questionnaire that is easy to use in
practice.11
|
Although attacks of migraine may start at any age, the incidence peaks
in early to mid-adolescence. In the United States and Western
Europe, the one-year prevalence of migraine is 11 percent overall: 6
percent among men and 15 to 18 percent among women.12,13,14
The median frequency of attacks is 1.5 per month, and the median duration
of an attack is 24 hours; at least 10 percent of patients have
weekly attacks, and 20 percent have attacks lasting two to three
days.12
Thus, 5 percent of the general population have at least 18 days of
migraine per year, and at least 1 percent — that is, more than 2.5
million persons in North America — have at least 1 day of migraine
per week. The lifetime prevalence of migraine is at least 18
percent,13
although among older subjects the figures are deflated by recall
bias. In the United States, most patients with migraine have not
seen a physician for headache during the previous year, have never
received a medical diagnosis of migraine, and use over-the-counter
medications to the exclusion of prescription drugs.15 A
recent survey by the World Health Organization (WHO) rates severe
migraine, along with quadriplegia, psychosis, and dementia, as one
of the most disabling chronic disorders.16
This ranking suggests that in the judgment of the WHO, a day with
severe migraine is as disabling as a day with quadriplegia.
Pathophysiology
Migraine is best understood as a primary disorder of the brain.17
It is a form of neurovascular headache: a disorder in which neural
events result in the dilation of blood vessels, which, in turn,
results in pain and further nerve activation.18
Migraine is not caused by a primary vascular event. Migraine attacks
are episodic and vary within and among patients. We may best explain
this variability by considering the basic biologic problem in
migraine to be the dysfunction of an ion channel in the aminergic brain-stem
nuclei that normally modulates sensory input and exerts neural
influences on cranial vessels.17
In patients with familial hemiplegic migraine, missense mutations
in the ![]() 1 subunit of
the voltage-gated P/Q-type calcium channel have been identified.19
It is possible that other ion-channel mutations contribute to
migraine without aura, since it is primarily cases of migraine with
aura that have been linked to the familial-hemiplegic-migraine locus.20
It thus seems possible that the aura of migraine is separate from
the headache,21
with aura susceptibility genes as its determinant22;
the pain and associated features of migraine itself may be determined
by another gene or genes.
1 subunit of
the voltage-gated P/Q-type calcium channel have been identified.19
It is possible that other ion-channel mutations contribute to
migraine without aura, since it is primarily cases of migraine with
aura that have been linked to the familial-hemiplegic-migraine locus.20
It thus seems possible that the aura of migraine is separate from
the headache,21
with aura susceptibility genes as its determinant22;
the pain and associated features of migraine itself may be determined
by another gene or genes.
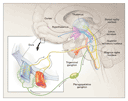
Migraine and the Brain
As noted above, migraine probably results from a dysfunction of
brain-stem or diencephalic nuclei that are involved in the sensory —
particularly nociceptive — modulation of craniovascular afferents. Activation
in the brain stem during attacks of migraine has been detected with
the use of positron-emission tomography.23,24
Moreover, the aura of migraine is likely to be the human counterpart
of the animal phenomenon of Leão's spreading depression.25
Aura is characterized by a wave of oligemia that passes across the
cortex26,27,28,29
at the characteristically slow rate of 2 to 6 mm per minute.30 A
short phase of hyperemia precedes this oligemia31
and is likely to be a correlate of such symptoms as flashing, jagged
lights. Oligemia is a response to depressed neuronal function and is
still clearly present when the headache starts.29,32
These findings, together with direct evidence that the local oxygen
supply is more than adequate,33
make the notion that migraine is simply a vascular headache untenable
(Figure 1).
|
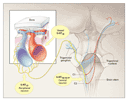
Pain Mechanisms
We do not completely understand the pathogenesis of pain in migraine,
but three key factors merit consideration: the cranial blood
vessels, the trigeminal innervation of the vessels, and the reflex
connections of the trigeminal system with the cranial parasympathetic
outflow. The substance of the brain is largely insensate; pain can
be generated by large cranial vessels,35
proximal intracranial vessels,36,37
or by the dura mater.38,39,40
These vessels are innervated by branches of the ophthalmic division of
the trigeminal nerve,40
whereas the structures of the posterior fossa are innervated by
branches of the C2 nerve roots.41
In nonhuman primates, stimulation of vascular afferents leads
to the activation of neurons in the superficial layers of the trigeminal
nucleus caudalis in the region of the cervicomedullary junction and
the superficial layers of the dorsal horns of the C1 and C2 levels
of the spinal cord42,43 —
the trigeminocervical complex. Similarly, stimulation of branches of
C2 activates neurons in the same regions of the brain.44,45,46
The involvement of the ophthalmic division of the trigeminal nerve
and the overlap with structures innervated by C2 explain the common
distribution of migraine pain over the frontal and temporal regions,
as well as the involvement of parietal, occipital, and high cervical
regions by what is, in essence, referred pain.
Peripheral trigeminal activation in migraine is evidenced by the
release of calcitonin-gene–related peptide, a vasodilator,47
but the mechanism of the generation of pain is not clear. Studies in
animals suggest that the pain may be caused by a sterile neurogenic
inflammatory process in the dura mater,48
but this mechanism has no clearly demonstrated correlate in humans.49
The pain may be a combination of an altered perception — as a
result of peripheral or central sensitization — of craniovascular
input that is not usually painful34
and the activation of a feed-forward neurovascular dilator mechanism
that is functionally specific for the first (ophthalmic) division of
the trigeminal nerve.50
Drug Therapy
Approaches to treating migraine can be divided into
nonpharmacologic therapies and pharmacologic therapies.
Nonpharmacologic therapies include education of the patient about
the disorder, its mechanisms, approaches to treatment, and changes
in lifestyle involved in the avoidance of triggers of migraine. In
patients with migraine, the brain does not seem to tolerate the
peaks and troughs of life well. Thus, regular sleep, regular meals,
exercise, avoidance of peaks of stress and troughs of relaxation,
and avoidance of dietary triggers can be helpful. The crucial
message is that the patient should aim for a certain regularity of
habits, rather than adhere to a long list of prohibitions of foods
and activities. What cannot be known is the sensitivity of the brain
to such triggers at any given time. This uncertainty leaves many
patients frustrated by the fact that the same manipulations intended
to avoid triggering migraine will lead to different outcomes on
different days. It can be helpful to explain the nature of this
variability to patients. An evidence-based review of nonpharmacologic approaches
to treatment of migraine was recently published.51
Drugs for the treatment of migraine can be divided into drugs
that are taken daily whether or not headache is present to reduce the
frequency and severity of attacks52
and drugs that are taken to treat attacks as they arise. Treatments
for attacks can be further divided into nonspecific and
migraine-specific treatments. Nonspecific treatments, such as
aspirin, acetaminophen, nonsteroidal antiinflammatory drugs,
opiates, and combination analgesics, are used to treat a wide range
of pain disorders. Specific treatments,7
including ergotamine, dihydroergotamine, and the triptans, are effective
for treating neurovascular headaches, such as migraine and cluster
headache, but not for treating other types of pain, such as pure
tension-type headache53
or atypical facial pain.54
Given that there are responses to placebo in patients with migraine,55
that there is a significant rate of nonresponse to oral drugs, and
that triptans have not been studied systematically in patients with
such problems as subarachnoid hemorrhage or meningitis, triptans
should not be used as diagnostic testing agents in patients with
headache.
Preventive Therapy
The decision to start a preventive therapy in a patient with migraine
is best made collaboratively. On the basis of a combination of the
frequency, duration, severity, and tractability of acute attacks, as
well as the preference of the patient, a sensible selection can be
made. Patients who have attacks that are unresponsive to
acute-attack medications and that cause substantial disability are
candidates for preventive therapy. If attacks occur at least twice a
month, if the patient may be at risk for rebound headache, or if the
migraine diary kept by the patient reveals a clear trend toward an
increasing frequency of attacks, it is probably better to consider
prevention than to wait for the problem to become more troublesome.
It is not clear how preventive therapy works, although it seems
likely that it modifies the sensitivity of the brain that underlies
migraine.
In general, if headaches occur one to two days per month, there
is usually no need for preventive therapy; if they occur three to
four days per month, preventive therapy should be considered; if the
patient has five or more attacks per month, preventive therapy
should be considered seriously. The available options are listed in Table 3, and the
evidence regarding their use has been extensively reviewed.50
Often, the doses required to reduce the frequency of headache cause
marked and intolerable side effects. Each drug should be started at
a low dose, and the dose should be gradually increased to a
reasonable maximum; patients should be reminded that this approach
often entails some delay in achieving efficacy.
|
On average, about two thirds of the patients given any of the drugs
listed in Table 3
will have a 50 percent reduction in the frequency of headaches. It
is our practice to explain the side effects of these drugs and
engage the patient in the decision-making process. We avoid
methysergide, at least initially, because of its fibrotic
complications,58,59
and we carefully explain the teratogenicity of divalproex
(valproate).60
Treatment of Acute Attacks
Analgesic and Nonsteroidal
Antiinflammatory Drugs
In many patients, migraine responds well to simple treatment at
the time of an attack. There are several key features of the
successful use of such treatments, after the preference of the
patient and any contraindications have been taken into consideration.
The drug should be taken as soon as the headache component of the
attack is recognized.61
The dose of drug should be adequate; for example, 900 mg of aspirin,62,63
1000 mg of acetaminophen,64
500 to 1000 mg of naproxen,65
400 to 800 mg of ibuprofen,66
or appropriate doses of a combination of these drugs.67,68
The administration of antiemetic drugs or drugs that increase
gastric motility is likely to facilitate the absorption of the
primary drug and thus help to ameliorate the attack.63,69,70
Overuse of these drugs should be avoided; for example, intake should
be restricted to no more than two to three days a week, and a
headache diary should be kept and monitored for any escalation in
drug use. It is important to remember that the severity of migraine
attacks and their response to treatment may vary; patients may
therefore require only one drug for some attacks but several drugs
for more bothersome attacks.
As a rule, we avoid the use of opiates. These drugs seem to mask
the pain without suppressing the pathophysiologic mechanism of the
attack, often leaving the patient cognitively impaired. Their use
may lead to addiction, and for most patients, they offer no advantages
over more migraine-specific therapy.
Ergot Derivatives
The main advantages of the ergotamine and dihydroergotamine ergot
derivatives are their low cost and the long experience with their
use.71,72
The major disadvantages are their complex pharmacology, their
erratic pharmacokinetics, the lack of evidence regarding effective
doses, their potent and sustained generalized vasoconstrictor
effects, which are associated with adverse vascular events, and the
high risk of overuse syndromes and rebound headaches.72
The Triptans
In comparison with the ergot derivatives, the triptans (Table 4) have
distinct advantages — notably, selective pharmacology, simple and
consistent pharmacokinetics, evidence-based prescription instructions,
established efficacy based on well-designed controlled trials,
moderate side effects, and a well-established safety record.5,87
The most important disadvantages of the triptans are their higher
cost and the restrictions on their use in the presence of
cardiovascular disease.
|
Pharmacology
and Mechanisms of Action
The triptans are serotonin 5-HT1B/1D–receptor agonists.
They were discovered88
as a result of studies of serotonin and migraine89,90,91,92,93
that led to the identification of an atypical 5-HT receptor.
Activation of the novel receptor can close cranial arteriovenous
anastomoses,94
and its anatomical distribution is restricted in vivo.95
Seven major subclasses of 5-HT receptors — classes 1 through 7 — are
now recognized.96,97
The triptans all activate the 5-HT1B/1D receptor and, to
a lesser extent, the 5-HT1A or 5-HT1F receptor. It is
likely that the 5-HT1B/1D–agonist activity is the primary
mechanism of the therapeutic effects of these drugs, although a
therapeutic action at the 5-HT1F receptor has not been excluded.98
Exclusively 5-HT1D–mediated effects were studied with the
use of PNU142633,99
but the results were inconclusive.100
This compound, which has exclusively neural action, produced some
chest symptoms remarkably similar to those that occur with triptans.101
We define a triptan as a 5-HT1B/1D–receptor agonist.102
Triptans have three potential mechanisms of action: cranial vasoconstriction,88
peripheral neuronal inhibition,48
and inhibition of transmission through second-order neurons of the
trigeminocervical complex.102
Which mechanism is the most important is as yet unclear.103
These actions inhibit the effects of activated nociceptive trigeminal
afferents and, in this way, control acute attacks of migraine (Figure 2).
|
There are five triptans in routine clinical use: sumatriptan, naratriptan,
rizatriptan, zolmitriptan, and almotriptan. Eletriptan was recently
approved in Europe; frovatriptan is awaiting approval; and
donitriptan is in preclinical development.104
During migraine attacks, the oral absorption of many drugs is
delayed,69
so there may be an advantage to nonoral methods of administration,
such as the use of nasal sprays, inhalers, suppositories, or injections.
Most patients, however, prefer oral formulations,105
which account for 80 percent of all triptan prescriptions; we therefore
focus on the oral formulations. Sumatriptan is also available in
subcutaneous,106
rectal,107
and intrana-sal108
formulations; these will be discussed separately. The pharmacokinetic
properties of the triptans are summarized in Table 4.
Safety
and Tolerability
It is crucial to distinguish between safety and tolerability in
discussing studies of treatments for acute migraine. Tolerability refers
to the extent of medically unimportant but clinically irritating
side effects of drugs, such as tingling, flushing, and sensations of
pressure; safety is assessed on the basis of records of medically
important side effects. Because the latter type of effects may be
rare, safety is best assessed after large-scale clinical exposure.
The triptans differ from one another in terms of tolerability but
not in terms of safety. The most frequent side effects are tingling,
paresthesias, and sensations of warmth in the head, neck, chest, and
limbs; less frequent are dizziness, flushing, and neck pain or
stiffness. Triptans can constrict coronary arteries and may cause
chest symptoms, sometimes closely mimicking angina pectoris. Such
symptoms may cause alarm, so the cardiovascular issues warrant discussion.
When patients are warned about these symptoms, they rarely cause
problems.
In rare instances, however, triptan therapy has been associated
with myocardial infarction.87
There has thus been general concern about the safety of triptans.
This concern is supported by in vitro pharmacologic studies that
demonstrate the potential of the triptans to constrict the coronary
vessels of humans, although ergotamine and dihydroergotamine have a
more potent and longer-lasting effect than the triptans.109
It is clear from anatomical studies using antibodies selective for
human 5-HT1B or 5-HT1D receptors110
that 5-HT1B receptors are located primarily in the cranial
circulation but are also found in the coronary circulation.111
There have been relatively few reports of clinically important
myocardial ischemia or infarction, despite the now very substantial
human exposure to triptans, particularly to sumatriptan.87
However, all triptans are 5-HT1B agonists, and thus the
sensible contraindications of ischemic heart disease, uncontrolled
hypertension, and cerebrovascular disease apply to the entire class.
Meta-Analysis of Studies of Oral Triptan Therapy
Most trials of triptans have been similar in design112
and in the population of patients studied — factors that facilitate
meta-analysis. We recently performed such a meta-analysis, using data
from 24,089 patients in 53 controlled clinical trials of triptans.113,114
We subtracted the rate of response in the placebo group from that in
the active-drug group and used this difference, the therapeutic
gain, as another means of comparing the results of the trials.115
All doses of all drugs have been compared with the standard of
sumatriptan at a dose of 100 mg.
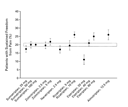
Improvement at Two Hours
The headache (pain) response at two hours was the primary end
point in nearly all trials of triptans. As compared with 100 mg
of sumatriptan, 10 mg of rizatriptan and 80 mg of eletriptan were
significantly more effective, whereas 2.5 mg of naratriptan, 20 mg
of eletriptan, and 2.5 mg of frovatriptan were less effective (data
were obtained from abstracts only) (Figure 3A).
|
Although the freedom from pain is the currently recommended primary
end point,116
it was a secondary end point in most trials. In terms of this end
point, 80 mg of eletriptan, 12.5 mg of almotriptan, and 10 mg of
rizatriptan were more effective than 100 mg of sumatriptan, whereas
25 mg of sumatriptan, 2.5 mg of naratriptan, and 20 mg of eletriptan
were less effective than 100 mg of sumatriptan (Figure 3B).
Sustained Freedom from Pain
The percentages of patients with sustained freedom from pain (freedom
from pain at 2 hours with no rescue medication and with no
recurrence of headache within 24 hours) are shown in Figure 4. These
rates were higher with 10 mg of rizatriptan, 80 mg of eletriptan,
and 12.5 mg of almotriptan than with 100 mg of sumatriptan, and
lower with 20 mg of eletriptan than with 100 mg of sumatriptan.
|
Intrapatient Consistency of Response
Efficacy in at least two out of three treated attacks can be considered
a reasonable estimate of consistency. Such consistency was found in
67 percent of patients given 100 mg of sumatriptan and 65 percent of
those given 50 mg of sumatriptan. The consistency of 10 mg of
rizatriptan was evaluated in a novel double-blind, crossover study
encompassing four attacks in each patient, with placebo given during
one randomly chosen attack in four of five groups of patients; the
fifth group received 10 mg of rizatriptan for each of four attacks.117
The unusual design, with the inclusion of placebo, makes it
difficult to compare this study with others, but it seems unlikely
that the inclusion of placebo would increase consistency. The rates
of consistency in the three attacks for which patients received
rizatriptan were the highest for any of the triptans; the rates of
response and freedom from pain were 86 percent and 48 percent,
respectively, in at least two out of three attacks and 60 percent
and 20 percent, respectively, in three of three attacks.
Tolerability
Differences among studies in the methods of collecting data on
adverse events and in the definitions of such events complicate comparisons.
The rates of adverse events with most triptans other than
sumatriptan overlap with those found with 100 mg of sumatriptan;
there were lower values for 2.5 mg of naratriptan and 12.5 mg of
almotriptan. The rates in the latter instances did not differ from
those found with placebo.
Direct Comparisons
In general, trials involving direct comparisons provide the optimal
comparison between drugs, although encapsulation of treatments,
selection bias, and population size may influence results. Some
direct comparisons between triptans have been conducted, and the
overall results of those to which we had access were consistent with
the results of the studies of single triptans.113
Comparisons between the main pharmacologic and clinical
characteristics of the new oral triptans and those of 100 mg of oral
sumatriptan are summarized in Table 5;
information on these characteristics is derived from a synthesis of
both types of studies.113
|
Parenteral Sumatriptan
Subcutaneous sumatriptan, at a dose of 6 mg, has the best
pharmacokinetic profile (time to maximal concentration, 10 minutes;
bioavailability, 96 percent),118
clinical efficacy (a response rate of 76 percent and a rate of freedom
from pain of 48 percent at 60 minutes after administration),106
and intrapatient consistency in multiple attacks (up to 90 percent).119
The main limitations are that patients must inject themselves and
that the incidence of adverse events is higher and their intensity
is greater than with oral sumatriptan. This adverse-event profile
may be related to the fixed 6-mg dose, among other factors, since 3
to 4 mg may suffice in many patients.120
Subcutaneous sumatriptan is also highly effective in the treatment
of acute attacks of cluster headache.121
The efficacy and tolerability profiles of rectal and intranasal sumatriptan
are very similar to those of the oral formulation.107,108
These formulations may be useful in patients with nausea. Intranasal
sumatriptan in a dose of 20 mg is the only triptan with demonstrated
efficacy in adolescents,122,123
in whom migraine attacks are usually of relatively short duration
and are associated with more prominent gastrointestinal symptoms and
a high rate of response to placebo.124,125
Selecting Initial Treatment for Acute Attacks
Migraine is a heterogeneous disorder, so the selection of initial
treatment for acute attacks depends on the severity and frequency of
the attacks, the associated symptoms, the preference of the patient,
and the history of treatment. In patients with little headache-related
disability, it is usually appropriate to initiate treatment with one
or more analgesic drugs and to escalate treatment as needed. In a
recent clinical trial, the probability of successful treatment with
aspirin and metoclopramide decreased as the severity of
headache-related disability increased. Among the most disabled 25
percent of patients (MIDAS grade IV126),
the attacks were controlled successfully with a combination of
aspirin and metoclopramide in only 26 percent of the patients.127
It is critical to establish realistic expectations and to advise
patients to seek follow-up care if treatment fails. In patients with
substantial disability, it is appropriate to prescribe a triptan
early in the course of treatment, in keeping with a stratified
approach to care.127
The Future of Migraine Treatment
Although the triptans represent an important advance, they are
ineffective in some patients. A crucial improvement would be a
treatment for acute attacks that had no vascular effects — in other
words, an antimigraine treatment with exclusively neural action.
If the hypothesis that neurogenic inflammation caused the pain
was correct, selective neuronally active compounds with peripheral action
should be effective.128
Unfortunately, antagonists of neurokinin-1 receptors (which mediate
the biologic actions of substance P),129,130,131,132,133
an endothelin antagonist,134
a neurosteroid,135
and two specific inhibitors of the extravasation of plasma protein
(CP122,288136
and 4991W93137)
have proved ineffective in clinical trials. The selective 5-HT1F–receptor
agonist LY33437098
was effective, but it may act on both peripheral and central
trigeminal targets.102
However, purely neural compounds do work. The ![]() -amino-3-hydroxy-5-methyl-4-isoxazole propionic acid–kainate
antagonist LY293558138
and GR79236, a selective adenosine-A1–receptor agonist,139,140
have proved effective in acute attacks of migraine. Another
nonvascular approach would be to block the effects of
calcitonin-gene–related peptide,47
and suitable compounds that do so are now available.141
Another approach is blockade of nitric oxide synthesis, which has
proved effective in one preliminary study.142
-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid–kainate
antagonist LY293558138
and GR79236, a selective adenosine-A1–receptor agonist,139,140
have proved effective in acute attacks of migraine. Another
nonvascular approach would be to block the effects of
calcitonin-gene–related peptide,47
and suitable compounds that do so are now available.141
Another approach is blockade of nitric oxide synthesis, which has
proved effective in one preliminary study.142
There are novel clinical-trial designs and end points that are
expected to reflect clinical practice more accurately. These end
points include efficacy over the course of multiple attacks (intrapatient
consistency), sustained freedom from pain over a 24-hour period, and
the preference of the patient. Furthermore, so-called ASAP (as soon
as possible) trials, in which patients are allowed to treat their
attacks as soon as they are sure migraine is developing, will better
reflect the nature of migraine treatment in real life.143
Finally, patients prefer not to have attacks at all. Current prophylactic
therapies for migraine are relatively nonspecific, their efficacy is
moderate, and they have substantial side effects.52
Studying the mechanisms involved in the onset of migraine23,24
and the predisposition to attacks144
is likely to lead to more specific, more efficacious, and
better-tolerated prophylactic drugs. We are very optimistic about
the future for persons with migraine.
Dr. Goadsby
is a Wellcome Senior Research Fellow.
Dr. Goadsby has received research grants from or served as a consultant
to Abbott, Allergan, Almirall Prodesfarma, AstraZeneca, Bristol-Myers
Squibb, Elan, Glaxo SmithKline, Merck, Ortho-McNeil, Pfizer,
Pharmacia, Pierre Fabre, and Vanguard.
Dr. Lipton has received research grants or served as a consultant
to Abbott, Allergan, American Home Products, AstraZeneca, Bristol-Myers
Squibb, Elan, Glaxo SmithKline, Johnson & Johnson, Merck, Pfizer,
Pharmacia, and Vanguard.
Dr. Ferrari has received research grants from or served as a consultant
to Abbott, Allergan, Almirall Prodesfarma, AstraZeneca, Elan, Glaxo
SmithKline, Merck, Pfizer, Pharmacia, and Pierre Fabre.
Source Information
From the Institute of Neurology, National Hospital for Neurology
and Neurosurgery, London (P.J.G.); the Departments of Neurology, Epidemiology,
and Social Medicine, Albert Einstein College of Medicine and the Montefiore
Headache Unit, New York (R.B.L.); Innovative Medical Research, Towson, Md.
(R.B.L.); and the Department of Neurology, Leiden University Medical Center,
Leiden, the Netherlands (M.D.F.).
Address reprint requests to Professor Goadsby at the Institute of
Neurology, Queen Sq., London WC1N 3BG, United Kingdom, or at [log in to unmask].
References
- Lance
JW, Goadsby PJ. Mechanism and management of headache. 6th ed. Boston:
Butterworth–Heinemann, 1998.
- Silberstein
SD, Lipton RB, Goadsby PJ. Headache in clinical practice. Oxford, England:
Isis Medical Media, 1998.
- Goadsby
PJ, Silberstein SD, eds. Headache. Vol. 17 of Blue books of practical
neurology. Boston: Butterworth–Heinemann, 1997.
- Lipton
RB, Diamond S, Reed M, Diamond ML, Stewart WF. Migraine diagnosis and
treatment: results from the American Migraine Study II. Headache
2001;41:638-645.
[Medline]
- Olesen
J, Tfelt-Hansen P, Welch KMA. The headaches. 2nd ed. Philadelphia:
Lippincott Williams & Wilkins, 2000.
- Headache
Classification Committee of the International Headache Society.
Classification and diagnostic criteria for headache disorders, cranial
neuralgias and facial pain. Cephalalgia 1998;8:Suppl 7:1-96.
- Goadsby
PJ, Olesen J. Diagnosis and management of migraine. BMJ 1996;312:1279-1283.
[Abstract/Full Text]
- Lance
JW. Headache and face pain. Med J Aust 2000;172:450-455.
[Medline]
- Rasmussen
BK, Olesen J. Migraine with aura and migraine without aura: an
epidemiological study. Cephalalgia 1992;12:221-228.
[Medline]
- Launer
LJ, Terwindt GM, Ferrari MD. The prevalence and characteristics of
migraine in a population-based cohort: the GEM study. Neurology
1999;53:537-542.
[Abstract/Full Text]
- Stewart
WF, Lipton RB, Kolodner K, Liberman J, Sawyer J. Reliability of the
Migraine Disability Assessment score in a population-based sample of
headache sufferers. Cephalalgia 1999;19:107-114.
[Medline]
- Stewart
WF, Lipton RB, Celentano DD, Reed ML. Prevalence of migraine headache in
the United States: relation to age, income, race and other
sociodemographic factors. JAMA 1992;267:64-69.
[Medline]
- Rasmussen
BK, Olesen J. Symptomatic and nonsymptomatic headaches in a general
population. Neurology 1992;42:1225-1231.
[Abstract]
- Steiner
TJ, Stewart WF, Kolodner K, Liberman J, Lipton RB. Epidemiology of
migraine in England. Cephalalgia 1999;19:305-306.abstract
- Lipton
RB, Stewart WF, von Korff M. Burden of migraine: societal costs and
therapeutic opportunities. Neurology 1997;48:Suppl 3:S4-S9.
[Abstract]
- Menken
M, Munsat TL, Toole JF. The Global Burden of Disease Study: implications
for neurology. Arch Neurol 2000;57:418-420.
[Medline]
- Goadsby
PJ. Pathophysiology of headache. In: Silberstein SD, Lipton RB, Dalessio
DJ, eds. Wolff's headache and other head pain. 7th ed. Oxford, England:
Oxford University Press, 2001:57-72.
- May A,
Goadsby PJ. The trigeminovascular system in humans: pathophysiologic
implications for primary headache syndromes of the neural influences on
the cerebral circulation. J Cereb Blood Flow Metab 1999;19:115-127.
[Medline]
- Ophoff
RA, Terwindt GM, Vergouwe MN, et al. Familial hemiplegic migraine and
episodic ataxia type-2 are caused by mutations in the Ca2+
channel gene CACNL1A4. Cell 1996;87:543-552.
[Medline]
- Terwindt
GM, Ophoff RA, van Eijk R, et al. Involvement of the CACNA1A gene
containing region on 19p13 in migraine with and without aura. Neurology
2001;56:1028-1032.
[Abstract/Full Text]
- Gowers
WR. A manual of diseases of the nervous system. Philadelphia: P.
Blakiston, 1888.
- Goadsby
PJ. Migraine, aura, and cortical spreading depression: why are we still
talking about it? Ann Neurol 2001;49:4-6.
[Medline]
- Weiller
C, May A, Limmroth V, et al. Brain stem activation in spontaneous human
migraine attacks. Nat Med 1995;1:658-660.
[Medline]
- Bahra A,
Matharu MS, Buchel C, Frackowiak RSJ, Goadsby PJ. Brainstem activation
specific to migraine headache. Lancet 2001;357:1016-1017.
[Medline]
- Leão
AAP. Spreading depression of activity in the cerebral cortex. J
Neurophysiol 1944;7:359-390.
- Olesen
J, Larsen B, Lauritzen M. Focal hyperemia followed by spreading oligemia
and impaired activation of rCBF in classic migraine. Ann Neurol
1981;9:344-352.
[Medline]
- Woods
RP, Iacoboni M, Mazziotta JC. Bilateral spreading cerebral hypoperfusion
during spontaneous migraine headache. N Engl J Med 1994;331:1689-1692.
[Full Text]
- Sanchez
del Rio M, Bakker D, Wu O, et al. Perfusion weighted imaging during
migraine: spontaneous visual aura and headache. Cephalalgia
1999;19:701-707.
[Medline]
- Cutrer
FM, Sorensen AG, Weisskoff RM, et al. Perfusion-weighted imaging defects
during spontaneous migrainous aura. Ann Neurol 1998;43:25-31.
[Medline]
- Lauritzen
M. Pathophysiology of the migraine aura: the spreading depression theory.
Brain 1994;117:199-210.
[Abstract]
- Hadjikhani
N, Sanchez Del Rio M, Wu O, et al. Mechanisms of migraine aura revealed by
functional MRI in human visual cortex. Proc Natl Acad Sci U S A
2001;98:4687-4692.
[Abstract/Full Text]
- Olesen
J, Friberg L, Olsen TS, et al. Timing and topography of cerebral blood
flow, aura, and headache during migraine attacks. Ann Neurol
1990;28:791-798.
[Medline]
- Cao Y,
Welch KMA, Aurora S, Vikingstad EM. Functional MRI-BOLD of visually
triggered headache in patients with migraine. Arch Neurol 1999;56:548-554.
[Medline]
- Burstein
R, Yarnitsky D, Goor-Aryeh I, Ransil BJ, Bajwa ZH. An association between
migraine and cutaneous allodynia. Ann Neurol 2000;47:614-624.
[Medline]
- McNaughton
FL, Feindel WH. Innervation of intracranial structures: a reappraisal. In:
Rose FC, ed. Physiological aspects of clinical neurology. Oxford, England:
Blackwell Scientific, 1977:279-93.
- Martins
IP, Baeta E, Paiva T, Campos J, Gomes L. Headaches during intracranial
endovascular procedures: a possible model of vascular headache. Headache
1993;33:227-233.
[Medline]
- Nichols
FT III, Mawad M, Mohr JP, Hilal S, Adams RJ. Focal headache during balloon
inflation in the vertebral and basilar arteries. Headache 1993;33:87-89.
[Medline]
- Cushing
H. The sensory distribution of the fifth cranial nerve. Bull Johns Hopkins
Hosp 1904;15:213-32.
- Penfield
W, McNaughton F. Dural headache and innervation of the dura mater. Arch
Neurol Psychiatry 1940;44:43-75.
- Feindel
W, Penfield W, McNaughton F. The tentorial nerves and localization of
intracranial pain in man. Neurology 1960;10:555-563.
- Arbab
MA-R, Wiklund L, Svendgaard NA. Origin and distribution of cerebral
vascular innervation from superior cervical, trigeminal and spinal ganglia
investigated with retrograde and anterograde WGA-HRP tracing in the rat.
Neuroscience 1986;19:695-708.
[Medline]
- Hoskin
KL, Zagami A, Goadsby PJ. Stimulation of the middle meningeal artery leads
to bilateral Fos expression in the trigeminocervical nucleus: a
comparative study of monkey and cat. J Anat 1999;194:579-588.
[Medline]
- Goadsby
PJ, Hoskin KL. The distribution of trigeminovascular afferents in the
nonhuman primate brain Macaca nemestrina: a c-fos immunocytochemical
study. J Anat 1997;190:367-375.
[Medline]
- Kerr
FWL. A mechanism to account for frontal headache in cases of
posterior-fossa tumors. J Neurosurg 1961;18:605-609.
- Goadsby
PJ, Knight YE, Hoskin KL. Stimulation of the greater occipital nerve
increases metabolic activity in the trigeminal nucleus caudalis and
cervical dorsal horn of the cat. Pain 1997;73:23-28.
[Medline]
- Bartsch
T, Goadsby PJ. Stimulation of the greater occipital nerve (GON) enhances
responses of dural responsive convergent neurons in the trigemino-cervical
complex in the rat. Cephalalgia 2001;21:401-402.abstract
- Goadsby
PJ, Edvinsson L, Ekman R. Vasoactive peptide release in the extracerebral
circulation of humans during migraine headache. Ann Neurol 1990;28:183-187.
[Medline]
- Moskowitz
MA, Cutrer FM. Sumatriptan: a receptor-targeted treatment for migraine.
Annu Rev Med 1993;44:145-154.
[Medline]
- May A,
Shepheard S, Wessing A, Hargreaves RJ, Goadsby PJ, Diener HC. Retinal
plasma extravasation in animals but not in humans: implications for the
pathophysiology of migraine. Brain 1998;121:1231-1237.
[Abstract]
- May A,
Buchel C, Turner R, Goadsby PJ. Magnetic resonance angiography in facial
and other pain: neurovascular mechanisms of trigeminal sensation. J Cereb
Blood Flow Metab 2001;21:1171-1176.
[Medline]
- Silberstein
SD, Rosenberg J. Multispecialty consensus on diagnosis and treatment of
headache. Neurology 2000;54:1553-1553.
[Full Text]
- Welch
KMA. Drug therapy of migraine. N Engl J Med 1993;329:1476-1483.
[Full Text]
- Lipton
RB, Stewart WF, Cady R, et al. 2000 Wolfe Award: sumatriptan for the range
of headaches in migraine sufferers: results of the Spectrum Study.
Headache 2000;40:783-791.
[Medline]
- Harrison
SD, Balawi SA, Feinmann C, Harris M. Atypical facial pain: a double-blind
placebo-controlled crossover pilot study of subcutaneous sumatriptan. Eur
Neuropsychopharmacol 1997;7:83-88.
[Medline]
- Jhee SS,
Salazar DE, Ford NF, Fulmor IE, Sramek JJ, Cutler NR. Monitoring of acute
migraine attacks: placebo response and safety data. Headache 1998;38:35-38.
[Medline]
- Mathew
NT, Rapoport A, Saper J, et al. Efficacy of gabapentin in migraine
prophylaxis. Headache 2001;41:119-128.
[Medline]
- Potter
DL, Hart DE, Calder CS, Storey JR. A double-blind, randomized,
placebo-controlled, parallel study to determine the efficacy of topiramate
in the prophylactic treatment of migraine. Neurology 2000;54:Suppl
3:A15-A15.abstract
- Graham
JR, Suby HI, LeCompte PR, Sadowsky NL. Fibrotic disorders associated with
methysergide therapy for headache. N Engl J Med 1966;274:359-368.
[Medline]
- Graham
JR. Cardiac and pulmonary fibrosis during methysergide therapy for
headache. Am J Med Sci 1967;254:1-12.
[Medline]
- Silberstein
SD. Divalproex sodium in headache: literature review and clinical
guidelines. Headache 1996;36:547-555.
[Medline]
- Cady RK,
Sheftell F, Lipton RB, et al. Effect of early intervention with
sumatriptan on migraine pain: retrospective analyses of data from three
clinical trials. Clin Ther 2000;22:1035-1048.
[Medline]
- Tfelt-Hansen
P, Henry P, Mulder LJ, Scheldewaert RG, Schoenen J, Chazot G. The
effectiveness of combined oral lysine acetylsalicylate and metoclopramide
compared with oral sumatriptan for migraine. Lancet 1995;346:923-926.
[Medline]
- Tfelt-Hansen
P, Olesen J. Effervescent metoclopramide and aspirin (Migravess) versus
effervescent aspirin or placebo for migraine attacks: a double-blind
study. Cephalalgia 1984;4:107-111.
[Medline]
- Lipton
RB, Baggish JE, Stewart WF, Codispoti JR, Fu M. Efficacy and safety of
acetaminophen in the nonprescription treatment of migraine: results of a
randomized, double-blind, placebo-controlled, population-based study. Arch
Intern Med 2000;160:3486-3492.
[Medline]
- Welch
KMA. Naproxen sodium in the treatment of migraine. Cephalalgia
1986;6:Suppl 4:85-92.
[Medline]
- Kellstein
DE, Lipton RB, Geetha R, et al. Evaluation of a novel solubilized
formulation of ibuprofen in the treatment of migraine headache: a
randomized, double-blind, placebo-controlled, dose-ranging study.
Cephalalgia 2000;20:233-243.
[Medline]
- Lipton
RB, Stewart WF, Ryan RE Jr, Saper J, Silberstein S, Sheftell F. Efficacy
and safety of acetaminophen, aspirin, and caffeine in alleviating migraine
headache pain: three double-blind, randomized, placebo-controlled trials.
Arch Neurol 1998;55:210-217.
[Medline]
- Goldstein
J, Hoffman HD, Armellino JJ, et al. Treatment of severe, disabling
migraine attacks in an over-the-counter population of migraine sufferers:
results from three randomized, placebo-controlled studies of the
combination of acetaminophen, aspirin, and caffeine. Cephalalgia
1999;19:684-691.
[Medline]
- Volans
GN. Absorption of effervescent aspirin during migraine. Br Med J
1974;4:265-268.
[Medline]
- Cottrell
J, Mann SG, Hole J. A combination of ibuprofen lysine (IBL) and
domperidone maleate (DOM) in the acute treatment of migraine:a
double-blind study. Cephalalgia 2000;20:269-269.abstract
- Appropriate
use of ergotamine tartrate and dihydroergotamine in the treatment of
migraine and status migrainosus (summary statement): report of the Quality
Standards Subcommittee of the American Academy of Neurology. Neurology
1995;45:585-587.
[Medline]
- Tfelt-Hansen
P, Saxena PR, Dahlof C, et al. Ergotamine in the acute treatment of
migraine: a review and European consensus. Brain 2000;123:9-18.
[Abstract/Full Text]
- Plosker
GL, McTavish D. Sumatriptan: a reappraisal of its pharmacology and
therapeutic efficacy in the acute treatment of migraine and cluster
headache. Drugs 1994;47:622-651.
[Medline]
- Cabarrocas
X, Salvà M. Pharmacokinetic and metabolic data on almotriptan, a new
antimigraine drug. Cephalalgia 1997;17:421-421.abstract
- Cabarrocas
X, Jansat JM, Ferrer P, Luria X. Pharmacokinetics of oral almotriptan
during and outside a migraine attack. Cephalalgia 2000;20:417-418.abstract
- Milton
KA, Allen MJ, Abel S, et al. The safety, tolerability, pharmacokinetics
and pharmacodynamics of oral and intravenous eletriptan, a potent and
selective "5HT1D-like" receptor partial agonist.
Cephalalgia 1997;17:414-414.abstract
- Morgan
P, Rance D, James G, Mitchell R, Milton A. Comparative absorption and
elimination of eletriptan in rat, dog and human. Cephalalgia
1997;17:414-414.abstract
- Buchan
P, Ward C, Zeig S. Frovatriptan pharmacokinetics are unaffected during a
migraine attack. Cephalalgia 1999;19:365-365.abstract
- Buchan
P, Keywood C, Ward C. Pharmacokinetics of frovatriptan (VML251/SB 209509)
in healthy young and elderly male and female subjects. Cephalalgia
1998;18:410-410.abstract
- Kempsford
RD, Baille P, Fuseau E. Oral naratriptan tablets (2.5-10 mg) exhibit
dose-proportional pharmacokinetics. Cephalalgia 1997;17:408-408.abstract
- Fuseau
E, Baille P, Kempsford RD. A study to determine the absolute oral
bioavailability of naratriptan. Cephalalgia 1997;17:417-417.abstract
- Sciberras
DG, Polvino WJ, Gertz BJ, et al. Initial human experience with MK-462
(rizatriptan): a novel 5-HT1D agonist. Br J Clin Pharmacol
1997;43:49-54. [Erratum, Br J Clin Pharmacol 1997;43:450.]
[Medline]
- Lee Y,
Conroy JA, Stepanavage ME, et al. Pharmacokinetics and tolerability of
oral rizatriptan in healthy male and female volunteers. Br J Clin
Pharmacol 1999;47:373-378.
[Medline]
- Seaber
E, On N, Phillips S, Churchus R, Posner J, Rolan P. The tolerability and
pharmacokinetics of the novel antimigraine compound 311C90 in healthy male
volunteers. Br J Clin Pharmacol 1996;41:141-147.
[Medline]
- Thomsen
LL, Dixon R, Lassen LH, et al. 311C90 (zolmitriptan), a novel centrally
and peripheral acting oral 5-hydroxytryptamine-1D agonist: a comparison of
its absorption during a migraine attack and in a migraine-free period.
Cephalalgia 1996;16:270-275.
[Medline]
- Palmer
KJ, Spencer CM. Zolmitriptan. CNS Drugs 1997;7:468-78.
- Welch
KMA, Mathew NT, Stone P, Rosamond W, Saiers J, Gutterman D. Tolerability
of sumatriptan: clinical trials and post-marketing experience. Cephalalgia
2000;20:687-695. [Erratum, Cephalalgia 2001;21:164-5.]
[Medline]
- Humphrey
PPA, Feniuk W, Perren MJ, Beresford IJM, Skingle M, Whalley ET. Serotonin
and migraine. Ann N Y Acad Sci 1990;600:587-598.
[Medline]
- Sicuteri
F, Testi A, Anselmi B. Biochemical investigations in headache: increase in
hydroxyindoleacetic acid excretion during migraine attacks. Int Arch Allergy
Appl Immunol 1961;19:55-58.
- Curran
DA, Hinterberger H, Lance JW. Total plasma serotonin,5-hydroxyindoleacetic
acid and p-hydroxy-m-methoxymandelic acid excretion in normal and
migrainous subjects. Brain 1965;88:997-1010.
[Medline]
- Anthony
M, Hinterberger H, Lance JW. Plasma serotonin in migraine and stress. Arch
Neurol 1967;16:544-552.
[Medline]
- Kimball
RW, Friedman AP, Vallejo E. Effect of serotonin in migraine patients.
Neurology 1960;10:107-111.
- Lance
JW, Anthony M, Hinterberger H. The control of cranial arteries by humoral
mechanisms and its relation to the migraine syndrome. Headache
1967;7:93-102.
[Medline]
- Johnston
BM, Saxena PR. The effect of ergotamine on tissue blood flow and the
arteriovenous shunting of radioactive microspheres in the head. Br J
Pharmacol 1978;63:541-549.
[Medline]
- Feniuk
W, Humphrey PPA, Perren MJ. The selective carotid arterial vasoconstrictor
action of GR43175 in anaesthetized dogs. Br J Pharmacol 1989;96:83-90.
[Abstract]
- Hoyer D,
Clarke DE, Fozard JR, et al. International Union of Pharmacology
classification of receptors for 5-hydroxytryptamine (serotonin). Pharmacol
Rev 1994;46:157-203.
[Abstract]
- Hartig
PR, Hoyer D, Humphrey PPA, Martin GR. Alignment of receptor nomenclature
with the human genome: classification of 5-HT1B and 5-HT1D receptor
subtypes. Trends Pharmacol Sci 1996;17:103-105.
[Medline]
- Goldstein
DJ, Roon KI, Offen WW, et al. Selective serotonin 1F (5-HT1F)
receptor agonist LY334370 for acute migraine: a randomised controlled
trial. Lancet 2001;358:1230-1234.
[Medline]
- Pregenzer
JF, Alberts GL, Im WB, et al. Differential pharmacology between the
guinea-pig and the gorilla 5-HT1D receptor as probed with
isochromans (5-HT1D-selective ligands). Br J Pharmacol
1999;127:468-472.
[Abstract/Full Text]
- Gomez-Mancilla
B, Cutler NR, Leibowitz MT, et al. Safety and efficacy of PNU-142633, a
selective 5-HT1D agonist, in patients with acute migraine.
Cephalalgia 2001;21:727-732.
[Medline]
- Fleishaker
JC, Pearson LK, Knuth DW, et al. Pharmacokinetics and tolerability of a novel
5-HT1D agonist, PNU-142633F. Int J Clin Pharmacol Ther 1999;37:487-492.
[Medline]
- Goadsby
PJ. The pharmacology of headache. Prog Neurobiol 2000;62:509-525.
[Medline]
- Humphrey
PPA, Goadsby PJ. The mode of action of sumatriptan is vascular? A debate.
Cephalalgia 1994;14:401-410.
[Medline]
- John GW,
Perez M, Pawels PJ, Le Grand B, Verscheure Y, Colpaert FC. Donitriptan, a
unique high efficacy 5-HT1B/1D agonist: key features and acute
antimigraine potential. CNS Drug Rev 2000;6:278-89.
- Lipton
RB, Stewart WF. Acute migraine therapy: do doctors understand what
patients with migraine want from therapy? Headache 1999;39:Suppl 2:S20-S26.
- The
Subcutaneous Sumatriptan International Study Group. Treatment of migraine
attacks with sumatriptan. N Engl J Med 1991;325:316-321.
[Abstract]
- Tepper
SJ, Cochran A, Hobbs S, Woessner M. Sumatriptan suppositories for the acute
treatment of migraine. Int J Clin Pract 1998;52:31-35.
[Medline]
- Dahlof
C. Sumatriptan nasal spray in the acute treatment of migraine: a review of
clinical studies. Cephalalgia 1999;19:769-778.
[Medline]
- Maassen
VanDenBrink A, Reekers M, Bax WA, Ferrari MD, Saxena PR. Coronary
side-effect potential of current and prospective antimigraine drugs.
Circulation 1998;98:25-30.
[Abstract/Full Text]
- Smith D,
Shaw D, Hopkins R, et al. Development and characterisation of human 5-HT1B-
or 5HT1D-receptor specific antibodies as unique research tools. J Neurosci
Methods 1998;80:155-161.
[Medline]
- Longmore
J, Shaw D, Smith D, et al. Differential distribution of 5HT1D- and
5HT1B-immunoreactivity within the human trigemino-cerebrovascular system:
implications for the discovery of new antimigraine drugs. Cephalalgia
1997;17:833-842.
[Medline]
- Pilgrim
AJ. Methodology of clinical trials of sumatriptan in migraine and cluster
headache. Eur Neurol 1991;31:295-299.
[Medline]
- Ferrari
MD, Roon KI, Lipton RB, Goadsby PJ. Triptans (serotonin, 5-HT1B/1D
agonists) in acute migraine treatment: a meta-analysis of 53 trials.
Lancet 2001;358:1668-1675.
[Medline]
- Ferrari
MD, Goadsby PJ, Roon KI, Lipton RB. Triptans (serotonin, 5-HT1B/1D
agonists) in migraine: methods and detailed results of a meta-analysis of
53 trials. Cephalalgia (in press).
- McQuay
HJ, Moore RA. Using numerical results from systematic reviews in clinical
practice. Ann Intern Med 1997;126:712-720.
[Medline]
- Tfelt-Hansen
P, Block G, Dahlof C, et al. Guidelines for controlled trials of drugs in
migraine: second edition. Cephalalgia 2000;20:765-786.
[Medline]
- Kramer
MS, Matzura-Wolfe D, Polis A, et al. A placebo-controlled crossover study
of rizatriptan in the treatment of multiple migraine attacks. Neurology
1998;51:773-781.
[Abstract]
- Fowler
PA, Lacey LF, Thomas M, Keene ON, Tanner RJN, Baber NS. The clinical
pharmacology, pharmacokinetics and metabolism of sumatriptan. Eur Neurol
1991;31:291-294.
[Medline]
- Visser
WH, Vriend RH, Jaspers MW, Ferrari MD. Sumatriptan in clinical practice: a
2-year review of 453 migraine patients. Neurology 1996;47:46-51.
[Abstract]
- Visser
WH, Ferrari MD, Bayliss EM, Ludlow S, Pilgrim AJ, Subcutaneous Sumatriptan
International Study Group. Treatment of migraine attacks with subcutaneous
sumatriptan: first placebo-controlled study. Cephalalgia 1992;12:308-313.
[Medline]
- The
Sumatriptan Cluster Headache Study Group. Treatment of acute cluster
headache with sumatriptan. N Engl J Med 1991;325:322-326.
[Abstract]
- Ueberall
MA, Wenzel D. Intranasal sumatriptan for the acute treatment of migraine
in children. Neurology 1999;52:1507-1510.
[Abstract/Full Text]
- Winner
P, Saper JR, Nett R, Asgharnejad M, Laurenza A, Peykamian M. Sumatriptan
nasal spray in the acute treatment of migraine in adolescent migraineurs.
Pediatrics 1999;104:Suppl:694-695.abstract
- Korsgaard
AG. The tolerability, safety and efficacy of oral sumatriptan 50mmg and
100mg for the acute treatment of migraine in adolescents. Cephalalgia
1995;16:98-98.
- Rothner
A, Edwards K, Kerr L, DeBussey S, Asgharnejad M. Efficacy and safety of
naratriptan tablets in adolescent migraine. J Neurol Sci 1997;150:Suppl:S106-S106.abstract
- Lipton
RB, Stewart WF, Edmeads J, Sawyer J. Clinical utility of a new instrument
assessing migraine disability: the Migraine Disability Assessment (MIDAS)
score. Headache 1998;38:390-391.abstract
- Lipton
RB, Stewart WF, Stone AM, Lainez MJA, Sawyer JPC, Disability in Strategies
of Care Study Group. Stratified care vs step care strategies for migraine:
the Disability in Strategies of Care (DISC) Study: a randomized trial.
JAMA 2000;284:2599-2605.
[Medline]
- Moskowitz
MA. Neurogenic versus vascular mechanisms of sumatriptan and ergot
alkaloids in migraine. Trends Pharmacol Sci 1992;13:307-311.
[Medline]
- Diener
HC. Substance-P antagonist RPR 100893-201 is not effective in human
migraine attacks. In: Olesen J, Tfelt-Hansen P, eds. Proceedings of the
VIth International Headache Seminar. Philadelphia: Lippincott-Raven, 1996.
- Goldstein
DJ, Wang O, Saper JR, Stoltz R, Silberstein SD, Mathew NT. Ineffectiveness
of neurokinin-1 antagonist in acute migraine: a crossover study.
Cephalalgia 1997;17:785-790.
[Medline]
- Goldstein
DJ, Offen WW, Klein EG, et al. Lanepitant, an NK-1 antagonist, in migraine
prevention. Cephalalgia 2001;21:102-106.
[Medline]
- Norman
B, Panebianco D, Block GA. A placebo-controlled, in-clinic study to
explore the preliminary safety and efficacy of intravenous L-758,298 (a
prodrug of the NK1 receptor antagonist L-754,030) in the acute treatment
of migraine. Cephalalgia 1998;18:407-407.abstract
- Connor
HE, Bertin L, Gillies S, Beattie DT, Ward P, GR205171 Clinical Study
Group. Clinical evaluation of a novel, potent, CNS penetrating NK1
receptor antagonist in the acute treatment of migraine. Cephalalgia
1998;18:392-392.abstract
- May A,
Gijsman HJ, Wallnofer A, Jones R, Diener HC, Ferrari MD. Endothelin
antagonist bosentan blocks neurogenic inflammation, but is not effective
in aborting migraine attacks. Pain 1996;67:375-378.
[Medline]
- Data J,
Britch K, Westergaard N, et al. A double-blind study of ganaxolone in the
acute treatment of migraine headaches with or without an aura in
premenopausal females. Headache 1998;38:380-380.abstract
- Roon KI,
Olesen J, Diener HC, et al. No acute antimigraine efficacy of CP-122,288,
a highly potent inhibitor of neurogenic inflammation: results of two
randomized, double-blind, placebo-controlled clinical trials. Ann Neurol
2000;47:238-241.
[Medline]
- Earl NL,
McDonald SA, Lowy MT. Efficacy and tolerability of the neurogenic
inflammation inhibitor, 4991W93, in the acute treatment of migraine.
Cephalalgia 1999;19:357-357.abstract
- Ramadan
N, Sang C, Chappell A, et al. IV LY293558, an AMPA/KA receptor antagonist,
is effective in migraine. Cephalalgia 2001;21:267-268.abstract
- Goadsby
PJ, Hoskin KL, Storer RJ, Edvinsson L, Connor HE. Inhibitory effects of
adenosine A1 agonists on the cat trigeminovascular system:a new target for
anti-migraine drugs? Cephalalgia 2001;21:352-352.abstract
- Humphrey
PP, Bland-Ward PA, Carruthers AM, et al. Inhibition of trigeminal nociceptive
afferents by adenosine A1 receptor activation: a novel approach
towards the design of new anti-migraine compounds. Cephalalgia
2001;21:268-269.abstract
- Doods H,
Hallermayer G, Wu D, et al. Pharmacological profile of BIBN4096BS, the
first selective small molecule CGRP antagonist. Br J Pharmacol
2000;129:420-423.
[Abstract/Full Text]
- Lassen
LH, Ashina M, Christiansen I, Ulrich V, Olesen J. Nitric oxide synthase
inhibition in migraine. Lancet 1997;349:401-402.
[Medline]
- Cady RK,
Lipton RB, Hall C, Stewart WF, O'Quinn S, Gutterman D. Treatment of mild
headache in disabled migraine sufferers: results of the Spectrum Study.
Headache 2000;40:792-797.
[Medline]
- Ophoff
RA, Terwindt GM, Frants RR, Ferrari MD. P/Q-type Ca2+ channel
defects in migraine, ataxia and epilepsy. Trends Pharmacol Sci
1998;19:121-127.
[Medline]
Edward E.
Rylander, M.D.
Diplomat American
Board of Family Practice.
Diplomat American
Board of Palliative Medicine.